Introduction
Materials and Methods
Cultural conditions for the bacterial and fungal strains
Co-cultivation of B. subtilis PE7 and B. amyloliquefaciens N3S
Statistical analysis
Results
Growth patterns and changes in the pH values of the monocultures and co-culture of B. subtilis PE7 and B. amyloliquefaciens N3S
IAA production of B. subtilis PE7 and B. amyloliquefaciens N3S in the individual cultures and co-culture
Different inhibitory effects of the culture filtrates of the individual cultures and co-culture on mycelial growth of S. sclerotiorum
Effect of the culture filtrates on the hyphal morphology of S. sclerotiorum
Discussion
Conclusions
Introduction
The intensive use of agrochemicals in the agricultural fields has posed harmful environmental impacts including severe contamination of water and soil due to the accumulation of toxic residues, alteration in the soil pH and microbiota, and interruption of the soil structure and stability (Elumalai et al., 2025). An increase in public awareness prompts finding an urgent solution to mitigate the environmental risks caused by the agrochemicals. The application of microbial agents is an eco-friendly approach for not only plant disease control but also improved crop growth and yield, offering a sustainable alternative to agrochemicals. Diverse strains of bacterial and fungal genera including Bacillus, Pseudomonas, Paenibacillus, Streptomyces, Trichoderma have been widely used as biocontrol agents due to their capacity of controlling various phytopathogens and enhancing plant growth and productivity (Ayaz et al., 2023). Among these, Bacillus species are increasingly recognized for their potential in developing successful commercial bioproducts owing to their growth ability in inexpensive culture media, rapid growth and prolonged survival rate via endospore formation under extreme environmental conditions (Etesami et al., 2023; Herrmann et al., 2024). This endospore formation makes these species favorable for overcoming different challenges such as thermal and dehydration stresses during formulation, processing and storage of final products (Nemutanzhela et al., 2014; Stamenković-Stojanović et al., 2019; Ramlucken et al., 2021; Herrmann et al., 2024). Bacillus species synthesize a variety of bioactive compounds including bacteriocins, antibiotics, cyclic lipopeptides, cell wall degrading enzymes and volatile organic compounds (VOCs) in response to biotic stresses (Prasad et al., 2023). A group of cyclic lipopeptides such as iturins, surfactins and fengycins is a key weapon of Bacillus species for directly attacking the cell membrane of phytopathogens, thereby disturbing the function and integrity of the membrane (Ongena and Jacques, 2008; Zhang et al., 2022; Markelova and Chumak, 2025). The secretion of stress-relieving substances including phytohormones, exopolysaccharides, 1-aminocyclopropane-1- carboxylic acid (ACC) deaminase by these species is also impressive in mitigation of plant abiotic stresses such as salinity and drought (Etesami et al., 2023). Under these stressful environmental conditions, Bacillus species utilize their ability to develop a structure of sticky extracellular polymeric matrix called biofilm for their colonization and persistence in plant root and rhizosphere, thereby providing the available nutrients for direct plant growth and development through phytohormone production and solubilization of insoluble nutrients, and the synthesis of antifungal lipopeptides and VOCs (Ongena and Jacques, 2008; Fessia et al., 2022; Tsotetsi et al., 2022; de Oliveira-Paiva et al., 2024; Zhou et al., 2024). Moreover, their successful colonization in the rhizosphere and phyllosphere induces an efficient production of antimicrobial metabolites for effective biocontrol activity against the phytopathogens (Bais et al., 2004; Altaf et al., 2017; Fan et al., 2017).
A number of recent studies indicates that co-cultivation of two or more bacterial and fungal species provokes the synthesis of new active metabolites and is also more efficient in achieving diverse spectra of metabolites than the monocultures (Li et al., 2020; Kim et al., 2021; Peng et al., 2021; Selegato and Castro-Gamboa, 2023). Moreover, co-inoculation of two bacterial species elevates the plant growth promoting efficacy leading to a remarkable improvement on the growth quality and yield of maize, ginger and pea (Anwar et al., 2019; Gavilanes et al., 2020; Jabborova et al., 2022). Our previous studies reveal the endospore-forming B. subtilis PE7 as a plant growth promoting bacterium (PGPB) possessing multiple plant growth promoting (PGP) traits (Han et al., 2023a). Strain PE7 alleviates P-deficient stress and mediates growth promotion of melon plants resulting in an increase in the values of plant growth parameters and the nutrient contents (Han et al., 2024). Moreover, the soil drenching with the broth culture of strain PE7 remarkably promotes the growth of tomato plants in terms of greater shoot and root weights (Han et al., 2023b). Recent study demonstrates that the soil-originated Bacillus amyloliquefaciens AB22-1 showed not only strong antifungal activity against phytopathogens but also PGP effect on lettuce (Lee et al., 2025). In this study, strain PE7 was co-cultured with an antagonistic bacterium, B. amyloliquefaciens N3S to determine their agro-active metabolite production. B. amyloliquefaciens N3S was isolated from Doenjang, the Korean fermented soybean paste and it has strong antagonistic activity against several plant pathogenic fungi under in vitro conditions. Therefore, the present study aims to observe the growth rates of B. subtilis PE7 and B. amyloliquefaciens N3S as well as their performance on releasing IAA and antifungal metabolites in the co-culture compared to their respective monocultures.
Materials and Methods
Cultural conditions for the bacterial and fungal strains
The bacterial strains, Bacillus subtilis PE7 (KACC 92549P) and Bacillus amyloliquefaciens N3S, were used in the present study. Two bacterial strains were sub-cultured on tryptone soy agar (TSA) medium at 30°C for 2 days. Each single colony of B. subtilis PE7 and B. amyloliquefaciens N3S was inoculated again in each flask containing sterile tryptone soy broth (TSB) medium at 30°C for 1 day and used as the precultures. The fungal strain, Sclerotinia sclerotiorum (KACC 45771) was obtained from Korean Agricultural Culture Collection (KACC) and cultured on potato dextrose agar (PDA) medium at 25 ± 2°C for 3 days.
Co-cultivation of B. subtilis PE7 and B. amyloliquefaciens N3S
(i) Growth rateThe pre-cultures (500 µL) of strain PE7 and strain N3S were respectively inoculated in each flask containing 500 mL of a sterile medium (per liter: glucose 3 g, yeast extract 0.5 g, tryptophan 0.4 g, K2SO4 0.1 g, MgSO4 0.1 g, K2HPO4 0.4 g, KH2PO4 0.1 g, NaCl 0.1 g, urea 0.2 g, pH 7.2) and 250 µL of each strain was co-inoculated in a flask containing the same medium. The flasks were kept in a shaking incubator at 130 rpm and 30°C for 5 days. The broth cultures from the flasks were sampled at each incubation day for determination of IAA production and the pH values were measured. The cultures were then serially diluted and spread on sterile TSA medium. The plates were incubated at 30°C for 2 days and the cell numbers of each bacterial strain in terms of colony forming unit (CFU) were counted.
(ii) Determination of IAA production The broth cultures from each incubation day were centrifuged at 12,000 rpm and 4°C for 15 min. The resulting supernatant (1 mL) and Salkowski’s reagent (2 mL) was mixed and kept at room temperature for 30 min. Then, the IAA content in each sample was measured at an absorbance of 530 nm using a UV spectrophotometer (Shimadzu, Japan).
(iii) Evaluation of antifungal activity The samples of the 3- and 5-day-old monocultures and co-cultures were used to evaluate antifungal activity against S. sclerotiorum. The supernatants were obtained by centrifuging the broth cultures at 12,000 rpm and 4°C. A syringe filter (0.2 µm) was used to eliminate the remaining endospores in the supernatant. Then, the culture filtrate (CF) was mixed with the sterile-cool PDA medium to prepare the final CF concentrations of 10%, 30% and 50%. The PDA medium not blended with the CF was used as the control and triplicate plates were prepared for each CF concentration. A 5-mm-diameter mycelial plug of the freshly grown colony of S. sclerotiorum was inoculated at the center of each CF-supplemented and non-CF-supplemented plate. The plates were kept in an incubator at 25°C and the colony diameter of the fungal pathogen was measured at 2 days after incubation. The mycelial growth inhibition was calculated based on the equation (Jeong et al., 2024);
where Dc represents the colony diameter of S. sclerotiorum in the control plate and Dt represents the colony diameter of S. sclerotiorum in the CF-treated plate.
A small piece of mycelia from the CF-treated and non-CF-treated fungal colony was sampled and observed under a light microscope (Olympus, Japan) to examine the hyphal deformation affected by the CF.
Statistical analysis
The results of IAA production and antifungal activity of the CFs were analyzed by the analysis of variance (ANOVA) using the statistical analysis system (SAS, 9.4). The mean values were compared by the least significant difference (LSD) test at a significance level of 0.05.
Results
Growth patterns and changes in the pH values of the monocultures and co-culture of B. subtilis PE7 and B. amyloliquefaciens N3S
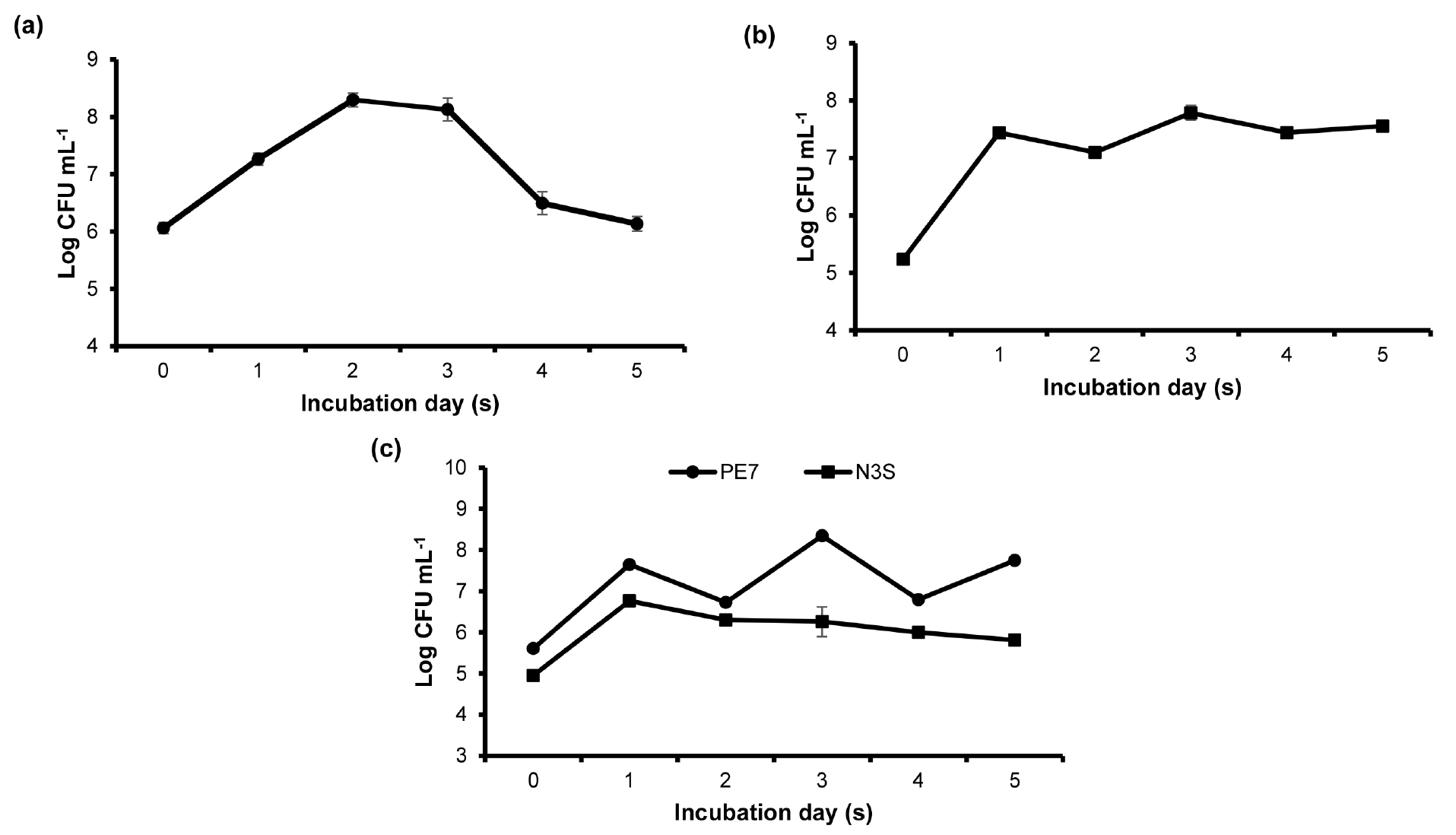
In the monoculture, B. subtilis PE7 gradually grew and the maximum growth (108 CFU mL-1) was observed at 2 days after incubation (DAI). Then, the growth slowly decreased at 3 DAI reaching the growth rate (106 CFU mL-1) at the final observation day (Fig. 1a). In contrast, a sharp increase of the growth was observed in the individual culture of B. amyloliquefaciens N3S at 1 DAI. However, the growth slightly dropped at 2 DAI and rose again at 3 DAI. From 4 DAI, there was no remarkable change in the growth until the end of the experimental period (Fig. 1b). In the co-culture, the growth patterns of the two bacterial strains were different. The growth of B. subtilis PE7 fluctuated during the observation period and the maximum growth rate was observed at 3 DAI. On the other hand, a rapid growth of B. amyloliquefaciens N3S was observed at 1 DAI and the growth was continuously stable until the end of the experimental period (Fig. 1c). In the co-culture experiment, two Bacillus species were distinguished by their distinct morphologies. B. subtilis PE7 possesses a flat, opaque and creamy-colored colony with irregular margin whereas B. amyloliquefaciens N3S colony is circular and off-white, and has a flower-like appearance with fringed margin. The colony size of strain N3S is smaller than that of strain PE7.
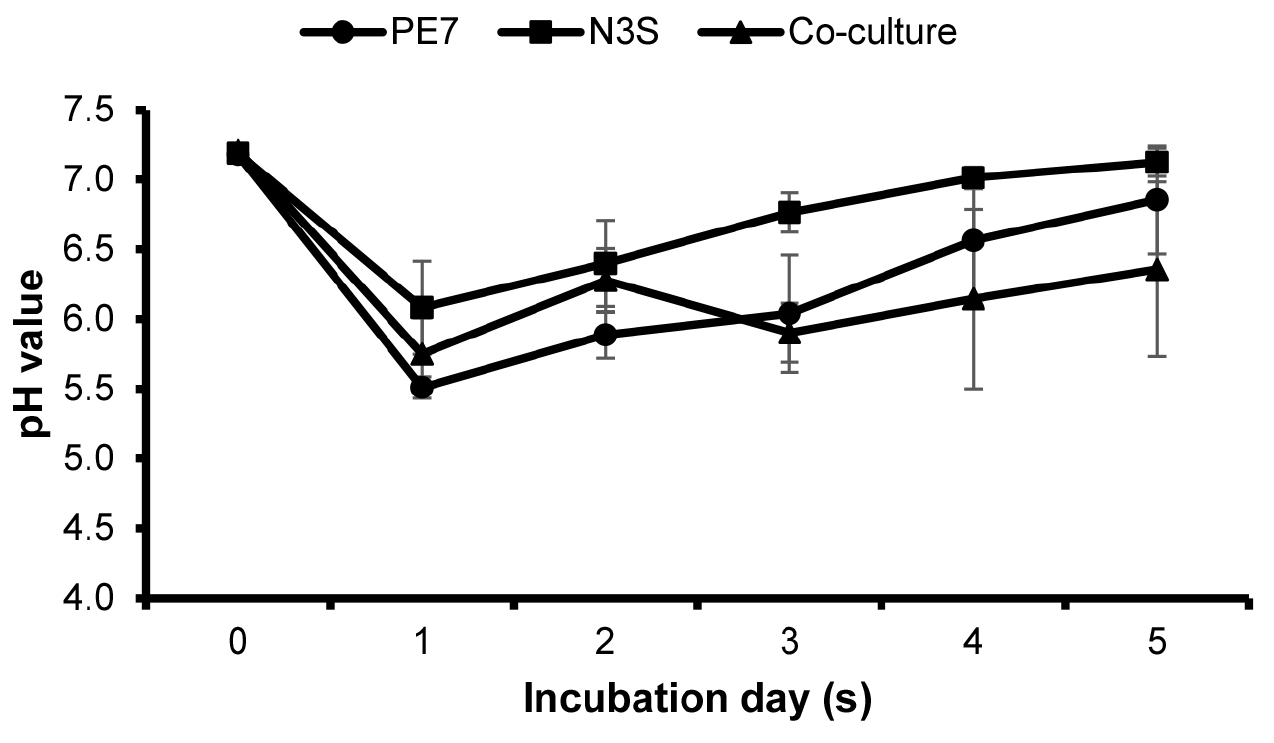
A similar trend of changes in the pH values was observed in the individual cultures and co-culture of B. subtilis PE7 and B. amyloliquefaciens N3S at the early observation days (Fig. 2). The pH values of all cultures dropped at 1 DAI and increased again at 2 DAI. In the individual cultures, a continuous increase in the pH values was observed until the end of the incubation period. However, the pH of the co-culture declined again at 3 DAI and rose again until the final observation day.
IAA production of B. subtilis PE7 and B. amyloliquefaciens N3S in the individual cultures and co-culture
A similar pattern of IAA secretion was observed in the individual culture of B. subtilis PE7 and the co-culture in which the IAA content decreased at 2 DAI and increased slowly until the end of the observation period (Fig. 3). In contrast, the IAA production of B. amyloliquefaciens N3S in the individual culture increased continuously from 2 DAI to 4 DAI and the production was stable at the final incubation day. The maximum IAA production with 9.94 µg mL-1 and 10.06 µg mL-1 in the monocultures of strain PE7 and strain N3S was observed at the final observation day.
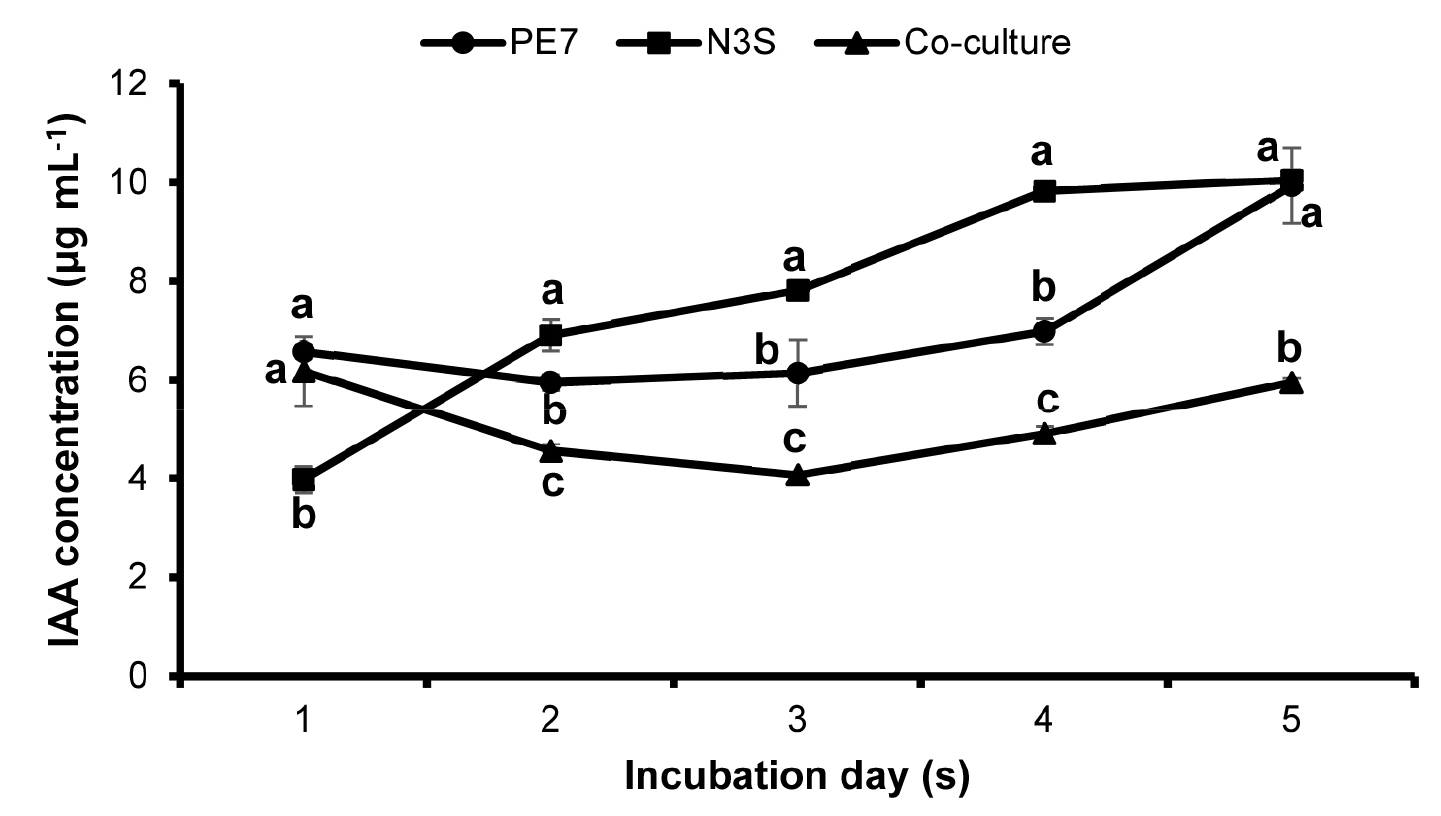
Fig. 3
The concentration of indole-3-acetic acid (IAA) produced by B. subtilis PE7 and B. amyloliquefaciens N3S in the monocultures and co-culture during the incubation period. The data represent mean ± standard deviation of three replicates. Different letters in the same incubation day indicate significant difference between each other as compared by the least significant difference (LSD) test at a significance level of 0.05.
Different inhibitory effects of the culture filtrates of the individual cultures and co-culture on mycelial growth of S. sclerotiorum
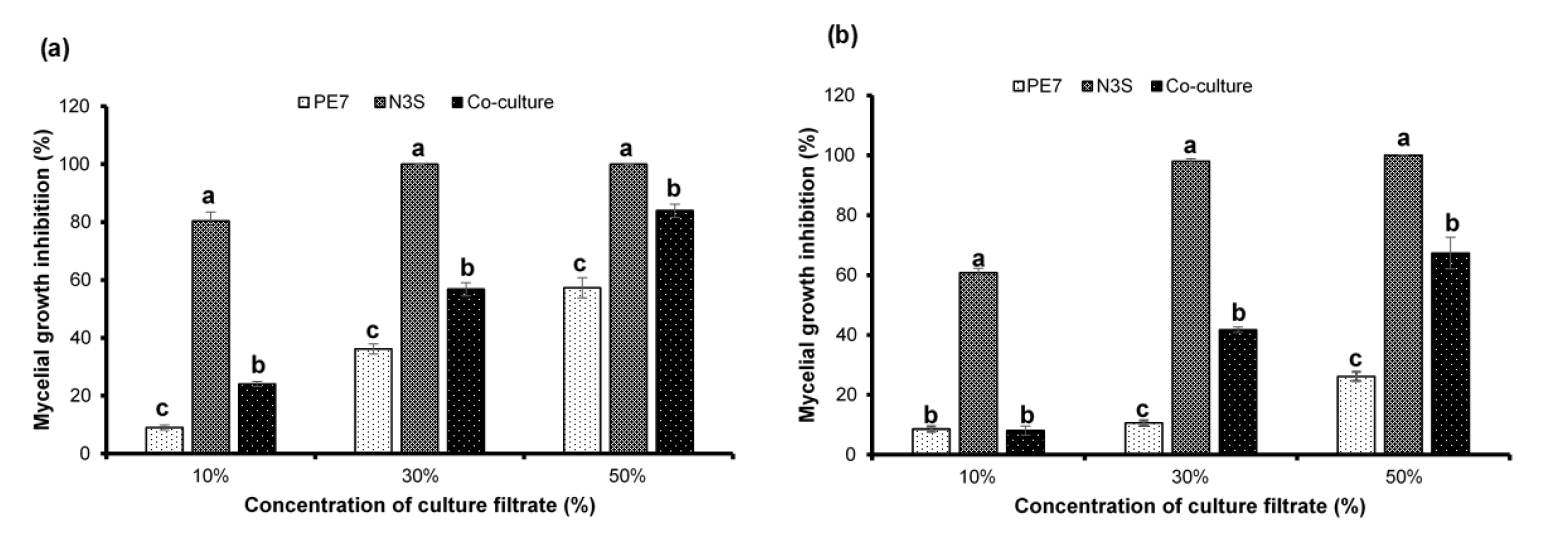
The inhibitory effect of the CFs on mycelial growth of S. sclerotiorum was concentration-dependent and the effect increased with the increased concentration of CF supplemented in the fungal growth medium. Moreover, the CFs of the 3-day-old cultures were more effective in reduced growth rate of S. sclerotiorum than those of the 5-day-old cultures (Fig. 4). The strongest antifungal activity was observed in treatments with the higher CF concentrations (30% and 50%) of both 3- and 5-day-old-individual cultures of B. amyloliquefaciens N3S, showing the absolute mycelial growth inhibition (100%) except an inhibition rate of 97.99% in treatment with 30% CF of the 5-day-old culture. Even the lowest CF concentration (10%) of the monoculture of strain N3S showed higher inhibitory effect than any CF concentration of the individual culture of B. subtilis PE7. Among the CFs of the monocultures and co-culture within each specific concentration, the CFs of strain PE7 were the least effective in reduction of mycelial growth showing the lowest inhibition rates. Interestingly, within every specific concentration, the CFs of the co-culture exhibited higher inhibition on mycelial growth than those of the individual culture of strain PE7.
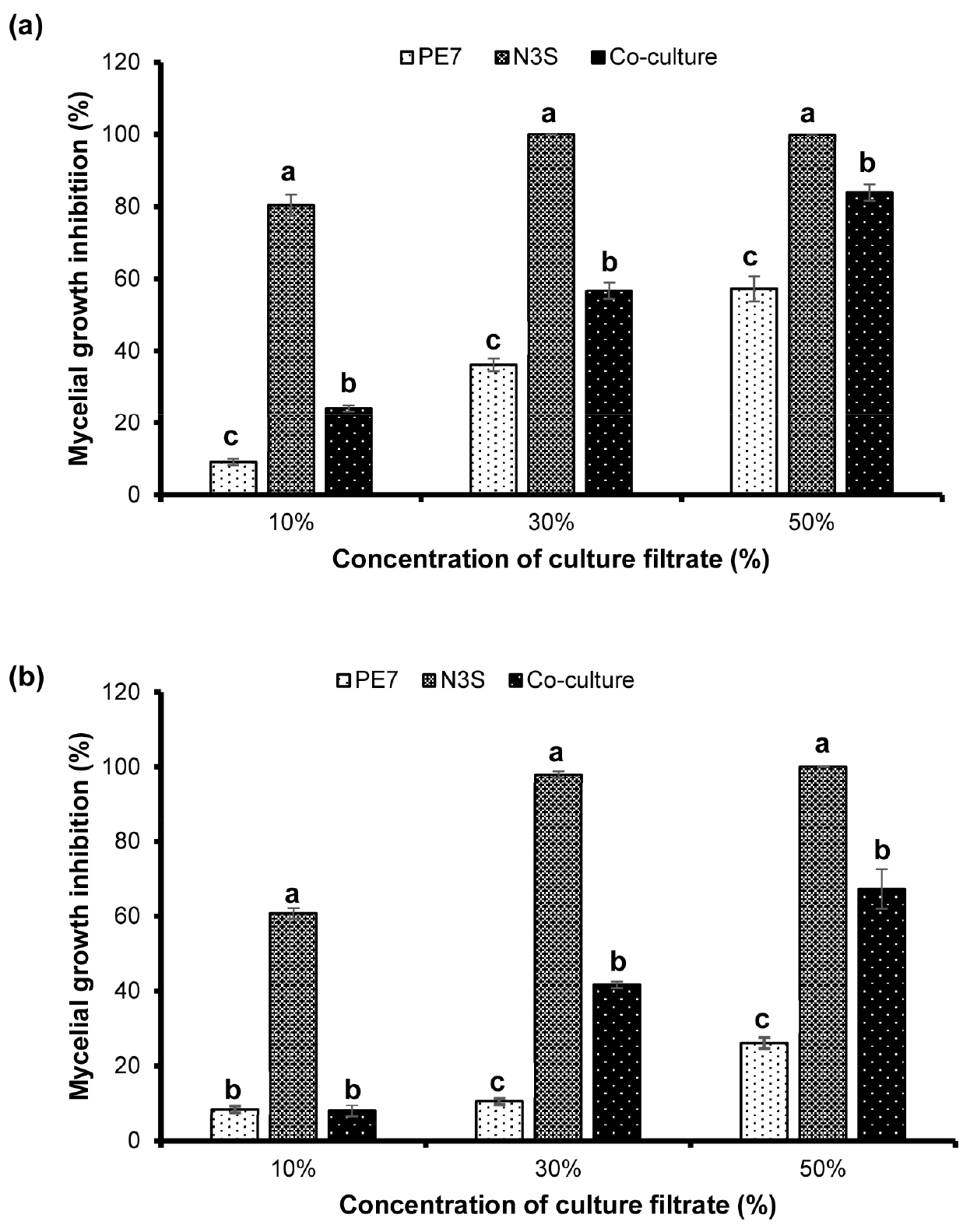
Fig. 4
Different levels of growth inhibition of S. sclerotiorum caused by different culture filtrate concentrations of 3-day-old (a) and 5-day-old (b) monocultures and co-culture of B. subtilis PE7 and B. amyloliquefaciens N3S. The data bar represents mean ± standard deviation of three replicates. Different letters on the bar graph within each culture filtrate concentration indicate significant difference between treatments as compared by the least significant difference (LSD) test at a significance level of 0.05.
Effect of the culture filtrates on the hyphal morphology of S. sclerotiorum
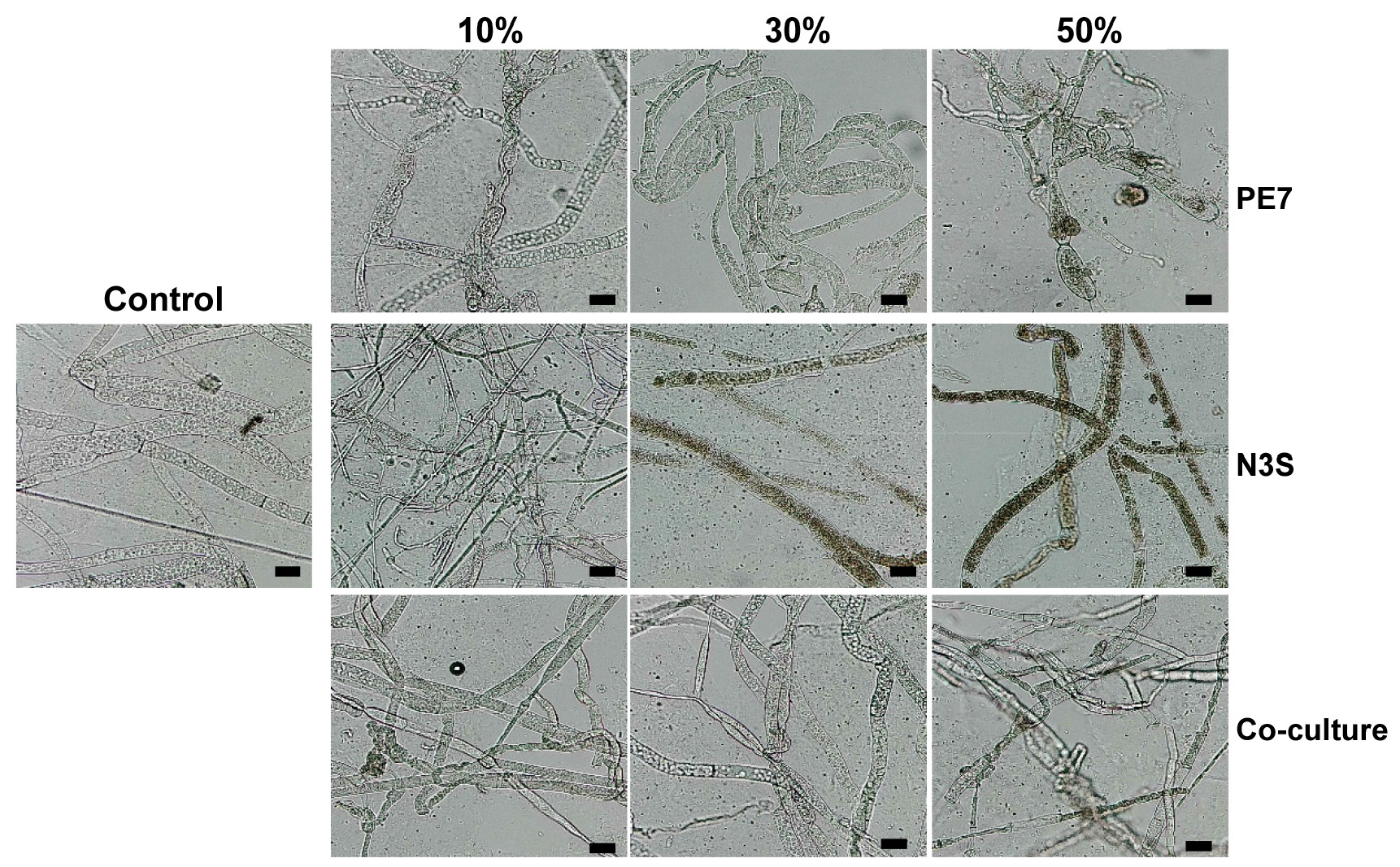
The exposure of the CFs to S. sclerotiorum induced different degrees and appearances of hyphal deformation (Fig. 5). Treatment with the CFs of the individual culture of B. subtilis PE7 caused abnormal changes to the hyphal morphology of S. sclerotiorum including coiling, swelling and irregular thinning with deflated shapes. The hyphae became distinctly thin and folded in treatment with the lowest CF concentration (10%) of the individual culture of B. amyloliquefaciens N3S whereas the exposure of the higher CF concentrations (30% and 50%) to S. sclerotiorum resulted in the collapse of the hyphae with massive production of melanin, and the internal contents of the hyphae were degraded. The exposure of the CFs of the co-culture to S. sclerotiorum induced morphological abnormalities of hyphae including knotting and swellings. Moreover, the distorted appearances were also observed in the hyphae treated by the highest CF concentration (50%) of the co-culture.
Discussion
The promising performance of Bacillus species under their monoculture conditions have been extensively studied for their ability to enhance plant growth and protect the plant from invasion of different phytopathogens, thereby improving plant health under biotic and abiotic stressed conditions (Shafi et al., 2017; Prasad et al., 2023). However, there are limited studies of the efficiency of these Bacillus species in a co-culture system for production of PGP and antimicrobial metabolites. In the present study, B. subtilis PE7 and B. amyloliquefaciens N3S was co-cultured to evaluate their growth rate as well as the level of the co-culture components such as IAA and antifungal metabolites compared to those of monocultures. During the experimental period, no growth extinction was observed although the growth rate of strain N3S in terms of cell numbers was lower than that of strain PE7. However, the growth of strain N3S seems to be slightly affected by the rapid growth of strain PE7 in the co-culture resulting in lower cell numbers compared to its monoculture on all observation days. As pH is one of the most important factors that substantially influences the growth and persistence of the bacterial species, modifying the environmental pH in the mixed culture mediates a positive interaction between different bacterial species leading to the cooperative growth (Ratzke and Gore, 2018). In our study, the growth rates of two Bacillus species indicate the stable coexistence although the pH values in the co-culture were lower than those of the monocultures at late observation days. Moreover, the IAA concentration in the co-culture was smaller than those of the monocultures in our study. Likewise, in another study by Bagheri et al. (2022), the IAA concentration released by a PGPB, Azospirillium oryzae NBT506 in the single culture was higher than that in the co-culture with Bacillusvelezensis UTB96. In different studies, the co-cultivation of two microbial strains induces the synthesis of new antimicrobial metabolites, and their interaction enhances the yield and diversity of these active substances resulting in better antimicrobial activity against pathogens (Yu et al., 2017; Li et al., 2020; Sun et al., 2022; Hamed et al., 2024). In our study, B. amyloliquefaciens N3S monoculture was the most effective in restriction of S. sclerotiorum growth whereas individual culturing of strain PE7 was ineffective in producing antifungal metabolites for mycelial growth inhibition of the fungal pathogen. Although the antifungal properties of the CFs of the co-culture was better than those of the single culture of PE7, the metabolite production of the co-culture was not much more efficient in inhibiting mycelial growth than that of the single culture of strain N3S. Therefore, further studies are necessary to explore the metabolic profile of the co-culture compared to those of strain PE7 and strain N3S monocultures, thereby revealing the possible mechanism and interaction between two Bacillus species in the co-culture. In addition, different studies reveal that metabolite production in the co-culture of diverse microbial strains was improved by optimizing medium components and other cultural parameters (Li et al., 2020; Liu et al., 2022). Therefore, to enhance the antifungal potency of the co-culture, the cultural conditions including medium composition and other factors as well as the ratio of the initial inoculum of two bacterial strains should be optimized.
In the present study, the mycelial growth inhibition as well as the development of hyphal deformations including swelling, distortion and bulging could be related to the action of diverse antifungal metabolites present in the CFs of the monocultures and co-culture. Bacillus-synthesized antimicrobial substances especially lipopeptides are well known for their ability to interact with the cell membrane of phytopathogenic fungi, thereby influencing membrane integrity and permeability via pore formation leading to the ultimate leakage of the intracellular components (Zhang et al., 2022; Markelova and Chumak, 2025). Several studies evinced severe alterations of the hyphal morphologies after exposure of Bacillus-lipopeptides to different fungal pathogens (Gong et al., 2015; Zhang and Sun, 2018; Xiao et al., 2021). Therefore, our study revealed the persistence of two Bacillus species, B. subtilis PE7 and B. amyloliquefaciens N3S in a coculture system through the release of IAA and antifungal metabolites for the growth inhibition of S. sclerotiorum. Further studies are required to examine and identify the major antifungal metabolites of the co-culture which are involved in antagonism against S. sclerotiorum in comparison with those of the corresponding monocultures. Moreover, the optimization of the growth medium and conditions is also required for efficient production of antifungal metabolites and other plant growth promoting substances in the coculture system.
Conclusions
The stable growth of B. subtilis PE7 and B. amyloliquefaciens N3S in a co-culture system along with the production of IAA and antifungal metabolites was revealed in this study. IAA production of each Bacillus strain in the monoculture was higher than that in the co-culture at most observation days. Although the bacterial CFs from both incubation periods were effective in controlling mycelial growth of S. sclerotiorum, a 3-day-incubation period induced higher antifungal metabolite production in both co-culture and monocultures than the 5-day-incubation period. The exposure of bacterial metabolites of the monocultures and co-culture to S. sclerotiorum resulted in hyphal deformations including coiling and thinning, and melanin production was also observed in the hyphae severely affected by the CFs. Therefore, the results of this study reveal the potential of co-inoculation of two Bacillus strains for their agro-active metabolite production. However, medium composition for enhanced production of these metabolites in the co-culture should be optimized as further studies.