Introduction
Soybean, Glycine max (L.) Merr, is a food crop and is considered the most important pulse crop in the world, providing proteins and edible oil to human and animals (Lenssen, 2013). South Korea relies on imports for most of its soybean requirement. Currently, 80,000 ha land is under soybean cultivation. Since 2003, the yield of soybean has been 130,000 ton accounting for a self-sufficiency rate of 32.5 and 8.4% for dietary and feed purposes, respectively. Thus, the soybean self-sufficiency rate in Korea is very low (Hong et al., 2011). Stable soybean production is significantly important from the industrial perspective because soybean is a major oil and feed crop. Lately, the demand for domestically grown soybean has been steadily increasing in South Korea owing to the recognition of its better quality and concerns over genetically modified imported soybean. To meet this demand, improvements in soybean cultivation techniques are desired because the soybean is one of the important crops which should be expansion of safe production base in dimension of the food security (Kim et al., 2010).
Of the various factors responsible for decreasing soybean production, diseases of the soybean plant have long been identified as the major limiting factor. Management of diseases as well as improvement in soil fertility is important for increasing the soybean productivity. In South Korea, yield and quality improvement of the soybean has become difficult because of the increase in damages caused by soybean stink bugs. These bean bugs cause damage to food crops as well as to sesame seeds, fruit trees, flowers, and medicinal plants (Paik et al., 2009; Kang et al., 2003). In South Korea, approximately 20 species of soybean stink bugs are known to damage soybean, especially, bean bugs (Riptortus clavatus), oriental green stink bugs (Nezara antennata), southern green stink bug (Nezara viridula), and hairy shield bug (Dolycoris baccarum), which occur at high frequency (Han and Choi, 1988). The stink bug, Riptortus clavatus Thunberg, is the most serious pest, which reduces the yield and quality of soybean (Lee et al., 1997; Ha et al., 1998).
Increased use of chemical pesticides for controlling these pests causes several negative effects such as the development of pathogen resistance to the applied agents, deterioration of human health, and environmental risk (Gerhardson, 2002). In addition, pesticide-free products are preferred by consumers. Use of functional microorgani-sms is being considered as an alternative for reducing the use of the chemicals in agriculture (De Weger et al., 1995; Gerhardson, 2002). Functional microorganisms have many potential uses as agents for enhancing plant growth and managing plant diseases (Glick, 1995; Weller, 1988). Plant growth-promoting bacteria are associated with many plant species and are commonly present in many environments (Kim et al., 2011). The active mechanisms of microbial biocontrol agents against plant pathogens include lytic activity (Woo et al., 2007), direct antibiosis (Ko et al., 2009), hyperparasitism (Lee et al., 2004), induction of resistance (Lee, 1997), and competition for space and nutrients (Jung et al., 2006). Some microorganisms act as plant growth promoting rhizobacteria (PGPR) that fix nitrogen (N), solubilize phosphorus (P), chelate iron (Fe), produce hormone-like substances, and degrade 1-aminocyclopropane-1-carboxylate (ACC) deaminase in soil (Jung et al., 2007; Kim et al., 2011).
Several studies have focused on the singular roles of microorganisms as biological pesticides or biofertilizers using in vitro experiments. However, the effects of application of functional microorganisms on soybean have rarely been studied. Therefore, in the present study, we determined the ability of Lysobacter antibioticus HS124 (gelatinase and chitinase producing microorganism; GCM), with plant growth promoting, and nematicidal and pesticidal activities (Ko et al., 2011; Lee et al., 2013; Kang et al., 2011), to promote soybean growth and control stink bugs in field trials by using field-onsite mass cultivation by farmers.
Materials and Methods
Fields, varieties of soybean, and GCM culture The experiments were conducted in the soybean fields of Sunchang-gun and Gimjae-si in Jeollabuk-do and Muan-gun in Jeollanam-do and Gwangju metropolitan city from June 2015 to November 2015. The soybean cultivars used were Haepum (Muan-gun), Daewon (Sunchang-gun), and Wooram (Gimjae-si and Gwangju). Seeding was performed on June 20, 2015 at Muan-gun, June 10, 2015 at Sunchang-gun, June 12, 2015 at Gimjae-si, and June 10, 2015 at Gwangju. Broth culture of Lysobacter antibioticus HS124 (GCM, Purne Korea) was prepared in a 500-L tank as follows: about 500 L water was filled in the tank, aerated using an air pump during the culture and adjusted to the temperature of 30°C with a heater. In addition, 0.5 L GCM inoculum, 0.01 kg gelatin (Geltec, Korea), and 0.5 kg crab shell powder (Purne, Korea) were added to the tank. After 3 days of culture, 2 L of highly enriched nutrient for microorganisms (Purne, Korea), 2.5-5 kg of sugar (Beksul, Korea), 2.5-5 kg of complex fertilizer (21-17-17, Dongbu Hannog, Korea) were put into the tank (Table 1) and cultured for 3-4 more days. The GCM broth was applied to each field at 500 L 0.3 ha-1 at 10-days intervals until final harvesting. The dilution of culture used for application included 1 portion of broth and 2-10 portions of water, as described later for individual fields. The GCM broth was applied 3 times to the Gimjae-si, Muan-gun, and Gwangju fields, whereas the Sunchang-gun field received 4 applications during the experimental periods.

†GCM: Gelatinase and chitinase producing microorganism
Cultivation method In Sunchang-gun, conventional practice treatment (CPT) as a control was done by planting seeds at a density of 2 seeds in an area of 36 × 100 cm2 soil, mulched by a black polythene film. The planting in GCM cultured broth treatment (GCMT) was at a density of 2 seeds in an area of 20 × 100 cm2, without mulch. In Gimjae-si, CPT and GCMT were performed at a planting density of 3 seeds in an area of 60 × 30 cm2. In Muan-gun, the planting density was 3 seeds in an area of 70 × 10 cm2 whereas in the Gwangju 1 field site, it was 2 seeds in an area of 90 × 28 cm2. In the Gwangju 2 field site, the density was 3 seeds in an area of 56 × 27 cm2 area.
Fertilization and management of diseases 15kg 10a-1 of complex fertilizer (N:P:K; 17-0-14) was used as a basal fertilizer in both CPT and GCMT in Sunchang-gun. In Gimjae-si, 10 kg 10a-1 of complex fertilizer (N:P:K:Mg, 12-6-6-4) was used as the basal fertilizer and 10 kg 10a-1 of complex fertilizer (N:P:K; 5-0-47) was used as an additional fertilizer in both CPT and GCMT. In Muan-gun, 40 kg 10a-1 of complex fertilizer (N:P:K; 21-17-17), 20 kg 10a-1 of KCl, and 20 kg 10a-1 of phosphorous fertilizer, such as fused phosphate, were used as the basal fertilizer, and 20 kg 10a-1 of urea and 20 kg 10a-1 of NK fertilizer were used as additional fertilizers in both CPT and GCMT. For controlling the stink bugs, each etofenprox and fenitrothion pesticide was applied once in both CPT and GCMT in Sunchang-gun; In Muan-gun, each etofenprox, fenitrothion, and chlorfenapyr pesticide was applied twice in both CPT and GCMT. In Gimjae-si and Gwangju 1 and 2 field sites, CPT received 500-1000 fold dilution of Eco-II (Purne, Korea), the organic sulfur (Purne, Korea), and organic surfactant (Purne, Korea) with 3 times. Whenever GCM was applied in GCMT at all fields, Eco-II, organic sulfur, and organic surfactant was mixed into GCM culture with 500-1000 times. Pheromone-baited traps (38 cm × 18 cm; B2M, South Korea) were used for assay of a bean bug (Riptortus clavatus Thunberg) and a brown-winged green bug (Plautia stali (Scott)) in CPT and GCMT. The traps were installed at a height of 60 cm above the ground separated by a minimum distance of 10 m between the traps in each field site. After one month, the bugs found inside the traps were collected from each field, taken to the laboratory where they were categorized by species, and were counted. This experiment was conducted for CPT and GCMT in all the field trials.
Soybean yield and yield components, and germination rate Analysis of soybean yield and yield components was conducted by the method described by Rural Development Administration (2003). Germination rate was determined by bean sprouters (ForUs, South Korea). Soybean seeds were placed in the bean sprouters and then kept at 25oC for 4 days with watering every 2 h a day. Germination was considered when length of the hypocotyls of the soybean seedlings was more than 5 mm.
Statistical analysis Experimental data were analyzed using standard one way analysis of variance (ANOVA) followed by Least Significant Difference (LSD) tests (P < 0.05) using the statistical analytical system (SAS, version 9.3; SAS Institute Inc, 2011).
Results and Discussion
Soybean yield and yield components, and germination rate Soybean yield and yield components are shown in Table 2. In the Muan-gun field, soybean yield of 336 kg in GCMT plants was 17% higher than the yield in CPT plants. The branches and pods plant-1, and seeds pods-1 in GCMT showed higher values than those in CPT, whereas the 100-seeds weight was same for both the treatments. In the Sunchang-gun field, soybean yield in GCMT plants was 20.3% higher than that in CPT plants. The yield components in GCMT showed higher values than in CPT. Although GCMT was not mulched, the soybean yield and yield components were higher than in CPT. However, the field in the Gimjae-si, soybean yield in GCMT plants was 10.6% lower than that in CPT plants and the pods plant-1 and seeds pod-1 in GCMT showed lower values than those in CPT. The reason for these results was not only the spraying of nitrogen fertilizer along with the application of GCM broth to soybean plants during the pollination period but also because of root rot diseases. In the Gwangju 1 field, soybean yield of 294 kg in GCMT plants was 19% higher than the 247 kg yield in CPT plants. The branches and pods plant-1 and seeds pod-1 in GCMT showed higher values than those in CPT. In the Gwangju 2 field, soybean yield of 274 kg in GCMT plants was 25.6% higher than the 218 kg yield in CPT plants. The branches and pods plant-1 and seeds pod-1 in GCMT showed higher values than those in CPT. The results showed that the soybean yield increased significantly (P < 0.05) by GCMT in Sunchang-gun, Gwangju 1 and Gwangju 2, while not significant in Muan-gun compared to CPT.
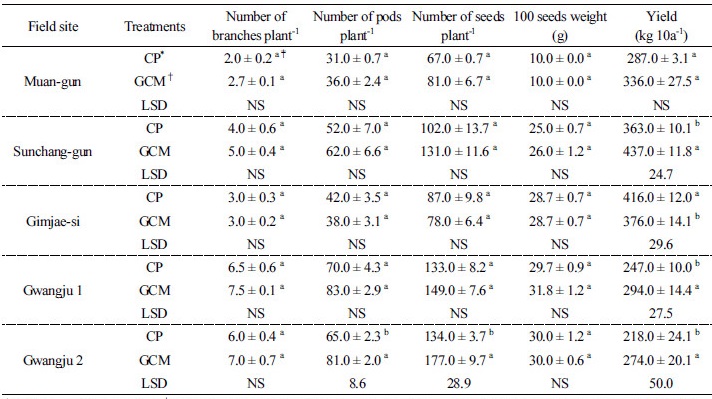
*CP: Conventional practice, †GCM: Gelatinase and chitinase producing microorganism; Calculated mean values are from three replicates ± SE (standard error); ‡Means within each variable followed by different letters are significantly different at P < 0.05 (LSD test). NS: Non-significant.
The increase in soybean yield may be related with the action of plant growth promoting metabolites in the GCM broth (Malik et al., 1997; Rodríguez and Fraga, 1999; Kim et al., 2008; Ryu et al., 2003). Furthermore, this might have resulted in the reduction of plant stress due to disease by the action of compounds for plant disease control produced by the GCM broth (Kloepper et al., 1980; Ko et al., 2009; Ko et al., 2011). The effect of plant growth promotion might be generated by GCMT which decomposing organic nitrogen into mineralized nitrogen in soil (Lee, 2004; Lim, 2005). Ko et al. (2011) also reported that the growth of pepper plants was increased by the treatment with L. antibioticus HS124 broth in comparison to the untreated control plants. However, we need to further verify whether GCM broth produces plant growth promoting compounds, such as phytohormones (Frankenberger and Arshad, 1995), as well as 4,5-dihydroxy-6-phenylhexanoic acid and volatile compounds (Farag et al., 2006) or not.
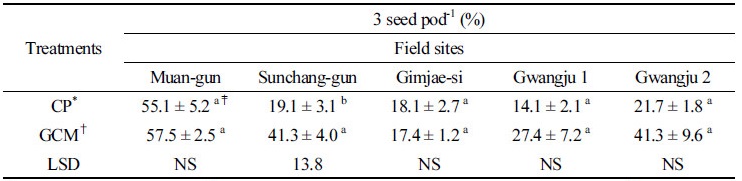
Table 3 shows that the percentages of 3 seed pod-1 in the GCMT plants from Sunchang-gun, Gwangju 1 and 2 were higher than those in CPT plants. However, the percentage of 3 seed pod-1 in the GCMT plants from Gimjae-si was slightly lower than that in CPT plants. The significant differences in 3 seed pod-1 were observed at Sunchang-gun, Gwangju 1, and Gwangju 2 in Table 3. These results suggest that GCMT might increase the formation of 3 seeded-pods in which resulted in the increased soybean yield in the former. Ratio of 3 seeds pod-1 is related to crop yield (Khare, 2011). Table 4 shows that although the increase in germination rates were observed at Muan-gun, Gimjae-si, Gwangju 1, and Gwangju 2, there was no significant difference except for Gwangju 2. Germination has relationship with the quality of seeds. According to Milosevic et al. (1992), seed size also affected seed germination.
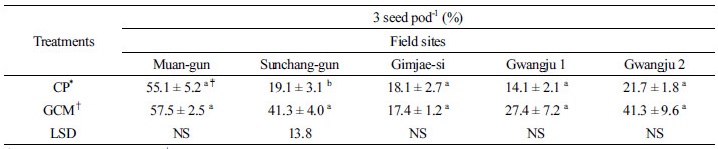
*CP: Conventional practice, †GCM: Gelatinase and chitinase producing microorganism; Calculated mean values are from three replicates ± SE (standard error); ‡Means within each variable followed by different letters are significantly different at P < 0.05 (LSD test). NS: Non-significant.
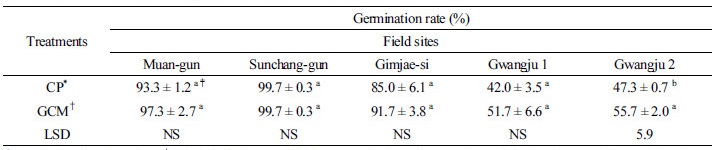
*CP: Conventional practice, †GCM: Gelatinase and chitinase producing microorganism; Calculated mean values are from three replicates ± SE (standard error); ‡Means within each variable followed by different letters are significantly different at P < 0.05 (LSD test). NS: Non-significant.
*CP: Conventional practice, †GCM: Gelatinase and chitinase producing microorganism; Calculated mean values are from three replicates ± SE (standard error); ‡Means within each variable followed by different letters are significantly different at P < 0.05 (LSD test). NS: Non-significant.
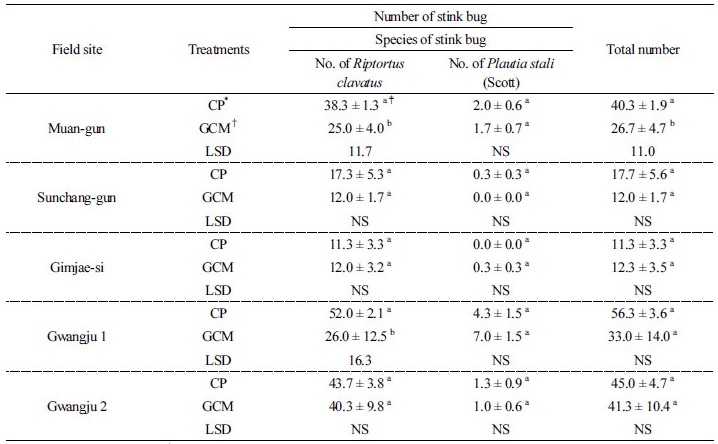
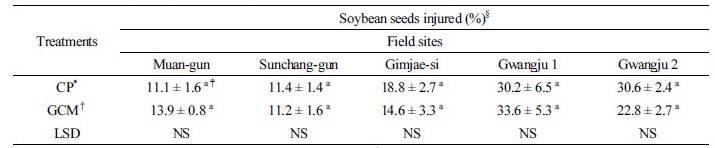
Occurrence and damage of stink bug As shown in Table 5, the occurrence of stink bug in GCMT was lower than that in CPT in all the fields except Gimjae-si, showing reductions of 47.5, 50.9, 70.6, and 8.9% in the Sunchang-gun, Muan-gun, Gwangju 1, and Gwangju 2 fields, respectively. The total number of stink bug in GCMT in Muan-gun significantly reduced as compared to that in CPT. These results might have been because of the effect of GCMT. As shown in Table 6, the percentage of damaged seeds in Sunchang-gun, Gimjae-si and Gwangju 2 in CPT was higher than in GCMT, whereas in Muan-gun and Gwangju 1, it in CPT was lower than that in GCMT. The percentage of damaged seeds by the stink bug in GCMT did not differ significantly (P < 0.05) to the all fields as compared to that in CPT. The result of seed damage by the stink bug in GCMT was inversely related to the occurrence of the stink bug in Muan-gun and Gwangju 1. Although the occurrence of stink bug decreased in Muan-gun, Sunchang-gun, Gwangju 1, and Gwangju 2 by GCMT, it did not coincide with the results of seed damage by the stink bug in GCMT plants. This anomalous observation about the stink bug control by GCMT in Muan-gun and Gwangju 1 remains unclear. However, in Sunchang-gun, and Gwangju 2, the control of stink bug by GCMT was effective based on the occurrence of stink bug and damaged seeds, although there are no statistically significant differences.
From this study, it could be concluded that the on-site application of culture broth of GCM, Lysobacter antibioticus HS124, on soybean fields could increase soybean productivity, as well as control the stink bug, although the results for them would be not interpreted as statistically significant in several field conditions. Further studies are required to clarify the actual mechanisms of GCM for more getting scientifically proofs in both promotion of soybean growth and suppression of stink bug by future application of GCM.
§Total seeds/seeds damaged × 100, *CP: Conventional practice, †GCM: Gelatinase and chitinase producing microorganism; Calculated mean values are from three replicates ± SE (standard error); ‡Means within each variable followed by different letters are significantly different at P < 0.05 (LSD test). NS: Non-significant.