Introduction
A portion of the veterinary antibiotics treated to livestock are generally excreted via feces and urine, and the antibiotics in animal manure may eventually enter soil and water environment and be taken up by vegetable crops (Kang et al., 2013; Tasho and Cho, 2016). In the Republic of Korea, 936 Mg of antibiotics were consumed in 2012 (APQA, 2013). Tetracyclines showed the highest consumption of 282 Mg, 30% of total amount. Seo et al. (2007) chose eight antibiotics including tetracyclines and tylosin to have a greater priority of environmental risk based on the consumption in Korea and the potential to reach soil and water environment.
It has been reported that the levels of antibiotics in water and soil near manure composting facilities in Korea were much higher than other countries (Lee et al., 2010; Ok et al., 2011; Awad et al., 2014). For instance, the maximum concentration of tetracycline in water, sediment and soil was 254.8 µg L-1, 32.5 µg kg-1, and 177.6 µg kg-1, respectively, in September (Awad et al., 2014). Kim et al. (2016) showed that tetracyclines could be the dominant residues in pig manures and reported that eight tetracycline resistance genes, tetG, tetH, tetK, tetY, tetO, tetS, tetW, and tetQ, were detected from agricultural soils in Korea.
Once the antibiotic residues enter the farmland, crops may obtain antibiotics in soils. Kumar et al. (2005) showed that chlortetracycline can be taken up by corn, green onion, and cabbage up to 17 ng g-1 on a fresh weight. Dolliver et al. (2007) reported that sulfamethazine was detected from lettuce, potato, and corn ranging from 0.1 to 1.2 mg kg-1 on a dry weight. Seo et al. (2010) also reported that lettuce, tomato, and hairy vetch could uptake chlortetracycline and tylosin up to 3.4 ng g-1 and 20.1 ng g-1 fresh weight, respectively.
Therefore, the antibiotics in animal byproducts need to be reduced or removed through composting or other agricultural practices prior to application of the livestock manure or slurry into agricultural fields. Many researchers have studied degradation of antibiotics during composting or storage of animal manure, for example, tylosin in swine, cattle, and chicken excreta (Teeter and Meyerhoff, 2003), chlortetracycline and oxytetracycline in beef manure (Arikan et al., 2009a, 2009b), chlortetracycline, sulfamethazine, and tylosin in pig manure (Kim et al., 2012a), chlortetracycline, tylosin, and monensin in horse manure (Kim et al., 2012b), chlortetracycline, tylosin, and sulfamethazine in chicken manure (Kwon et al., 2012), amprolium, lasalocid, monensin, and salinomycin in poultry litter and dairy manure (Arikan et al., 2016), sulfamethoxazole, sulfadimethoxine, and sulfamethazine in sludge (Yang et al., 2016), sulfadiazine, sulfathiazole, and sulfamethazine in swine manure (Chu et al., 2017), ciprofloxacin, enrofloxacin, lomefloxacin, norfloxacin, and sarafloxacin in broiler manure (Yang et al., 2017), and oxytetracycline in chicken manure (Ravindran and Mnkeni, 2017).
The objective of this study was to investigate the effect of aeration on degradation of tetracycline (TC) and tylosin (TYL) during storage of swine slurry.
Materials and Methods
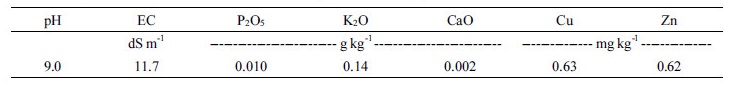
The swine slurry was obtained from a farm in Chuncheon, Korea, and its chemical characteristics are presented in Table 1. Two antibiotics, TC and TYL, were detected from the swine slurry used in the study with concentrations of 199 ng L-1 and 57 ng L-1, respectively. Treatments included aeration for 1, 3, and 6 hours per day in addition to no aeration as control. The aeration rate was 2.5 m3-air hr-1 m-3.
Samples for antibiotic analysis were taken on 9, 20, 28, 34, 42, 48, 55, and 62 days after aeration. Antibiotic levels in the swine slurry were determined using Enzyme-linked immunosorbent assay (ELISA) techniques (Aga et al., 2003; Kumar et al., 2004; Dolliver et al., 2008a; Seo et al., 2010). Samples were filtered through 0.45 µm syringe filter and the filtrate was used to analyze tylosin. For determination of tetracycline, samples were treated with ethylene diamine tetraacetic acid prior to filtration.
The levels of antibiotics in swine slurry were determined with ELISA kits (R-Biopharm AG, Darmstadt, Germany for tetracyclin and Tecna S.r.l., Trieste, Italy for tylosin). Briefly, sample or standard solution was added to wells coated with tetracycline-protein conjugate followed by adding antibody solution to the wells. Enzyme conjugate was added after removing any unbound antibody in a washing step and. Unbound enzyme-labeled antibodies were removed with a washing step followed by adding enzyme substrate and chromogen. Tetracycline levels were determined by measuring the absorbance at 450 nm after adding stop solution.
For TYL, the enzyme conjugate was added to compete with TYL in the sample for binding sites on the capture antibody after adding standards and samples to wells coated with high affinity capture antibody. Any unbound enzyme conjugate was removed through a series of washings followed by adding color-developing solution. The absorbance at 450 nm was measured after adding stop solution to stop the reaction.
A first-order kinetic model (Eq. 1) was used to express the degradation of TC and TYL in swine slurry.
dC/dt = -kC (Eq. 1)
where C is the concentration of the antibiotics and t and k is time (day) and the rate constant (day-1), respectively. The dissipation kinetic model (Eq. 2) can be derived from Eq. 1.
Ct = C0 × e-kt (Eq. 2)
where Ct and C0 is the measured concentration at a certain time t and the initial concentration of the antibiotics, respectively. Half-life of the antibiotics is the time to decrease the concentration to a half and can be calculated by Eq. 3 derived from Eq. 2.
t1/2 = ln2/k = 0.693/k (Eq. 3)
Results and Discussion
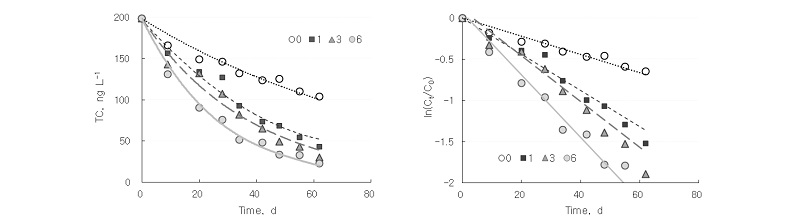
Aeration showed more rapid degradation of tetracycline. Over a period of 62 days, 78%, 85%, and 89% of initial tetracycline was dissipated by aeration for 1, 3, and 6 hours per day, respectively, compared with 48% under anaerobic condition (Fig. 1). Aeration for 1, 3, and 6 hours every day increased k for tetracycline from 0.011 day-1 under anaerobic condition to 0.022, 0.026, and 0.037 day-1, respectively (Table 2). A higher k value indicates faster dissipation. Aeration decreased half-life from 63.0 days for anaerobic storage to 31.5, 26.3, and 18.8 days for 1, 3, and 6 hours per day, respectively.
Table 2. Calculated parameters for fitting dissipation of tetracycline in swine slurry.

†mean ± standard deviation.
For composting of solid animal manure, decrease in level of antibiotics can be mainly through physical and chemical processes including sorption to organic matter rather than biotic processes (Arikan et al., 2009a; Kim et al., 2012a). Arikan et al. (2009a) reported that extractable chlortetracycline in the composted beef manure mixture and sterilized mixture decreased 99% and 98%, respectively at 55°C in 30 days. Yang et al. (2016) observed that adding spent mushroom compost to sludge enhanced degradation of sulfonamides compared with sludge alone. Kim et al. (2012a) suggested that organic materials produced from decomposition of pig manure and sawdust contributed to removal of tetracyclines. They concluded that organic matter provided additional binding sites for antibiotics during composting. Chu et al. (2017) reported that biological degradation material and moisture content were the major factors for biodegradation of sulphonamides during anaerobic composting of swine manure.
In the case of liquid swine slurry, biotic processes can be more effective than abiotic processes because of low organic matter and high water content. Yang et al. (2016) suggested that microbial degradation is a main process of the removal of sulphonamides in sludge. They reported that Acinetobacter and pseudomonas were main bacterial communities responsible for the degradation of sulphonamides. Qian et al. (2016) found that aerobic composting of cow manure increased antibiotic resistance genes such as tet (C), tet (X), sul1, sul 2, and intI1. Sarmah et al. (2006) and Dolliver et al. (2008b) reported that aerobic conditions and thermophilic temperatures rapidly reduced levels of antibiotics.
Degradation of antibiotics during composting of animal manure can be influenced by manure matrix and concentration (Arikan et al., 2016; Chu et al., 2017; Yang et al., 2017). Arikan et al. (2016) showed more rapid dissipation of monensin in dairy manure than in poultry litter. Chu et al. (2017) suggested that interactions between the initial concentration of antibiotics and temperature increased antibiotic degradation rates during anaerobic composting of swine manure. Yang et al. (2017) showed concentration-dependent and time-dependent patterns on removal of fluoroquinolone antibiotics during broiler manure composting.
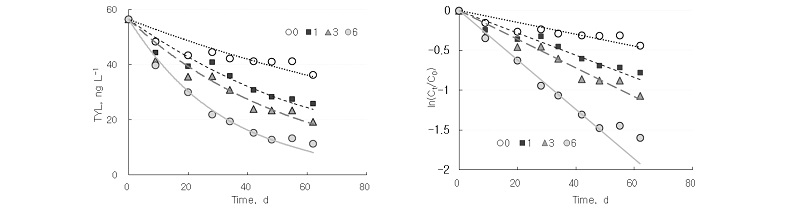
Tylosin showed slow degradation compared with tetracycline in this experiment. Under anaerobic condition, only 36% of initial tylosin was dissipated in 62 days. As seen with tetracycline, aeration enhanced the degradation rate of tylosin. Aeration for 1, 3, and 6 hours per day enhanced the dissipation to 54%, 66%, and 80%, respectively (Fig. 2). Aeration for 1, 3, and 6 hours per day increased k from 0.0074 day-1 to 0.014, 0.018, and 0.031 day-1, respectively (Table 3). Kim et al. (2012b) reported that the rate constant of tylosin for stockpiling horse manure was 0.030 day-1. Aeration for 1, 3, and 6 hours per day decreased half-life from 93.6 days to 49.5, 38.5, and 22.4 days, respectively. Halling-Sorensen et al. (2005) observed that half-life of tylosin in soil was 49-76 days. Kolz et al. (2005) showed more rapid dissipation of tylosin in pig manure under aerobic condition than under anaerobic condition.
Table 3. Calculated parameters for fitting dissipation of tylosin in swine slurry.
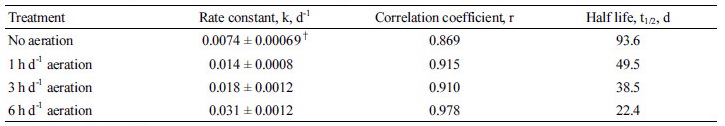
†mean ± standard deviation.
The results of the study suggested that aeration during storage of swine slurry for a short period of time could be a more efficient practice to decrease antibiotic levels prior to agricultural land application presumably through activity of aerobic microorganisms.
Conclusions
Degradation of tetracycline and tylosin in swine slurry was evaluated for aeration, one of the common management practices for liquid animal waste. Calculated half-life of tetracycline for aeration treatment ranged from 18.8 to 31.5 days compared with 63.0 days under anaerobic condition. For tylosin, the half-life decreased from 93.6 days without aeration to 22.4-49.5 days by aeration treatment. In the present experiment, aeration showed enhanced degradation of antibiotics in swine slurry compared with anaerobic storage.