Introduction
Plant growth promoting rhizobacteria can increase plant height and productivity due to their phytohormones synthesis, increasing availability of nutrients and mediating uptake of nutrients by the plants (Burd et al., 2000). Most of the interest has focus on non-symbiotic rhizobacterial strains, particularly Pseudomonas and Bacillus species. However, many still remain to be learned from the free living bacteria that have unique growth-enhancing effect on host plants (Bais et al., 2004; Ping and Boland 2004). Wu et al. (2005) demonstrated obviously the plant growth promoting effects of PGPR strains on different crops. Bacterial inoculants enhance plant growth, germination and seedling emergence (Lugtenberg et al., 2002). Salinity is abiotoc stress that substantially declines the yield of the crops by more than 50% (Bray, 2000). When salinity results from high level of NaCl, homeostasis of not only Na+ and Cl- but also K+ and Ca2+ is disordered (Serrano et al., 1999; Hasegawa et al., 2000; Rodriguez, 2000).High soil salinity is very serious factor limiting the agricultural production in a wide proportion over the world (Zhang and Hodson, 2001; Bybordi et al., 2010). Various environmental stresses such as water stress and high salt result in “stress ethylene” in which the ethylene is synthesized as a response to these stresses (Ciardi et al., 2000). This ethylene stress induces the symptoms of senescence in plant such as abscission and chlorosis which affect plant growth and survival (Glick, 2012). Some Rhizobia strains can produce the enzyme 1-aminocyclopropane-1-carboxylate (ACC) deaminase which hydrolyze the ACC which is the essential precursor for ethylene biosynthesis in plants (Ma et al., 2002). Therefore, ACC deaminase-producing PGPR may enhance plant growth by declining deleterious effects of stress ethylene. PGPR were found to tolerate salinity stress, which are often noxious to growth of important crop plants (Bacilio et al., 2004). After Honma and Shimomura (1978) discovered the enzyme 1-amino-cyclopropane-1-carboxylate (ACC) deaminase, Glick et al. (2007) showed that this is a common feature of many PGPB. Plant growth promoting rhizobacteria (PGPR) are comprised almost exclusively of free-living bacteria that dwell the rhizosphere of the plant (Kloepper et al., 1989). PGPR can enhance plant growth by various direct and indirect mechanisms (Glick, 1995; Gupta et al., 2000). Amongst of these mechanisms is improving plant stress tolerance to drought, salinity, and metal toxicity. Failure of germinations on saline soils is often due to high salt level in the planting zone as well as upward movement of soil solution and subsequent evaporation at the soil surface (Bybordi, 2010). The purpose of the present study was to evaluate the capability of some rhizobacteria strains to elevate the tolerance against salinity stress and to accelerate the early seed germination of Chinese cabbage Brassica rapa var. glabra.
Materials and Methods
Microorganisms and inoculums preparation Indole acetic acid (IAA) strongly producing bacteria were previously isolated from Panaxschinsen rhizosphere of Korean ginseng farm in Chuncheon city, Gangwon-do, South Korea. The strains were subcultured in Tryptic soy agar (TSA) medium pH 7.2. Bacterial strains were then preserved on (TSA) at 4°C (Noor et al., 2013). After two hours of growth in 5 mL of the nutrient broth as pre‒culture the OD of the culture broth was measured at 600 nm (OD600). One mL of the fresh grown spores was introduced into 250 mL new TSB broth medium supplemented with (5%) NaCl and incubated at 30°C for 48 h for activation.
NaCl stress effect on the seed germination of Chinese cabbage Plant seeds were surface sterilized by soaking in 70% ethanol for 1 min, then rinsed thoroughly in three treatments of sterile distilled water. Ten seeds were placed per Petri dish lined by sterilized filter paper (Whatman No. 1) wetted by 5 mL sterile solution of 0, 50, 100 and 150 mM NaCl. The experiment was conducted in three replicates. All Petri dishes of different treatments were incubated at 28°C under dark condition in plant growth chamber (DS 54 GLP, DASOL Scientific Co., Ltd., Korea). Germination progress was examined daily for 4 days. The number of germinated seeds was recorded every 24 h.
Inoculation of Chinese cabbage seeds The bacterial sus-pension was adjusted into 1×107 cfu mL-1 for each strain using UV-VIS Spectrophotometer (Hitachi U-2900), and optical density OD at 600 nm. Seeds were agitated at 150 rpm with its corresponding spore suspension for 2 h and at room temperature. Seeds were removed from the spore suspension and placed as 10 seeds/ plate. The seeds were landed on sterile filter (Whatman No. 1) which is humidified by 5 mL of sterile distilled water. The experiment was conducted in triplicate.
Inoculation of Chinese cabbage seeds under salinity stress The germination test of the seeds under different salinity levels of NaCl (50, 100 and 150 mM) was conducted in presence of either single or multi inoculated conditions. Seeds surface was sterilized and washed as mentioned above. Seeds were inoculated by soaking for 2h in 30 mL of its corresponding bacterial suspension (1×107cfu mL-1) with shaking conditions of 150 rpm. In case of mixed inoculation, 10 mL of each culture were participated. 30 mL sterile distilled water was served as control as well as other treatments. Seeds were filtrated using autoclaved gauze and placed exactly ten ones per Petri dish lined by sterile filter (Whatman No. 1). Salinity stress and humidity conditions were then provided by 5 mL sterile solution of 0, 50, 100 and 150 mM NaCl. The germination rate was calculated using Timson index (Khan and Ungar, 1985) of germination velocity.
Statistical analysis Data were run to analysis of variance (ANOVA). Significance at 5% level was tested by Least Significant Difference (LSD) using SAS program version 9.1 (SAS, 2009).
Results and Discussion
When plants are subject to a variety of stresses they often exhibit some symptoms which are known as ethylene syndrome. There is evidence that PGPR can interfere with ACC synthase, thus decline the concentration of the precursor of ethylene. Moreover, direct inoculation by phytohormones producing PGPR may affect plant metabolism. Many studies have shown that several soil bacteria, particularly the PGPR can produce cytokinins and gibberellins which are able to improve plant growth conditions (De Salamone et al., 2001). Hence, PGPR can stimulate plant tolerance to high salinity levels. In this work we investigated the effect of selected PGPR strains on the early germination of Chinese cabbage Brassica rapa var. glabra seeds.
PGP bacteria effect on seeds germination, radicle length, and plumule growth under different salinity levels Inoculation of A. chroococcum, Lactobacillus sp., and LAPmix could enhance the germination rate of Chinese cabbage seeds by 5% under salinity stress of 100 mM of NaCl. However, P. putida did not show increase in germination rate comparing to uninoculated seeds at the same salinity level. At salinity level of 150 mM of NaCl each of A. chroococcum, Lactobacillus sp., and LAPmix increased the germination rate by 5% rather than control. At salinity level of 200 mM of NaCl each of only Lactobacillus sp. and LAPmix increased the germination rate by 5% rather than control (Table 1 ). Mishra et al. (2010) demonstrated that the application of PGPR isolates signi-ficantly improved the percentage of seed germination under saline conditions. Under salinity stress, PGPR showed positive effect in plants evidenced by germination rate, tolerance of drought, yield and plant growth (Kokelis et al., 2006). Rhizo-bacteria have various mechanisms to survive in the rhizosphere under water stress and high salinity of soil (Miller and Woods 1996).
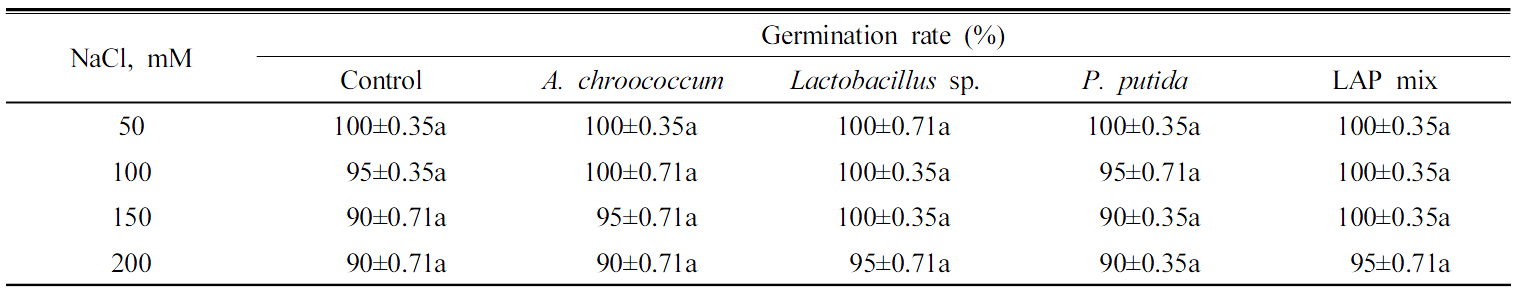
Under different salinity levels, inoculation of A. chroococcum showed remarkable enhancement to the radicle length of Chinese cabbage seedling where it showed 4.0, 1.2, and 1.0 times greater at treatments of 50, 100, and 150 mM of NaCl, respectively (Fig. 1 ). However, inoculation of A. chroococcum with 200 mM of NaCl increased the radicle length only by 0.3 cm. LAPmix enhanced the radicle length rather than the uninoculated control of the Chinese cabbage seedling by 2.0, 0.6, 1.0, and 0.8 cm under salinity stress of 50, 100, and 150 mM of NaCl, respectively. P. putidaand Lactobacillus sp. inoculations showed lower increase in the radicle length Chinese cabbage seedling (Table 2). IAA produced by bioferilizers enhances plant growth by increasing the number of root hairs and lateral roots (Okon and Kapulnik, 1986). PGPR stimulate plant growth via production of phytohormone, activation of phosphate solubilizing, suppression of deleterious organism, and promotion of the minerals nutrients uptake (Lalande et al., 1989).
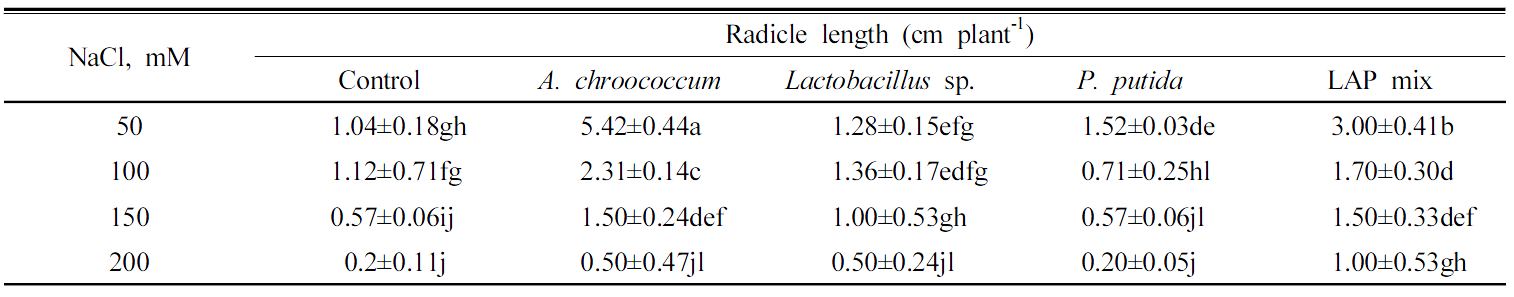
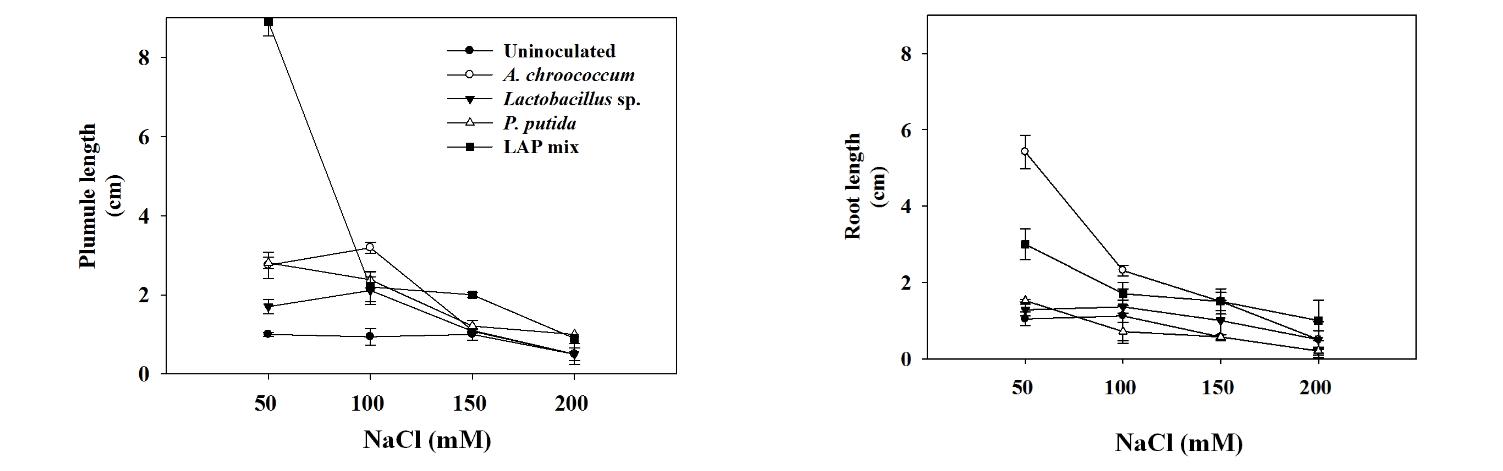
Fig. 1
Effect of PGPB inoculation on the early seed germination under salinity stress; a, shows the effect of the PGPR bacterial inoculation to enhance the salinity tolerance of the tested plants, represented in the plumule length, of Chinese cabbage Brassica rapa var. glabra; b, shows the effect of the PGPR bacterial inoculation to enhance the salinity tolerance of the tested plants, represented in the root length, of Chinese cabbage Brassica rapa var. glabra.
Inoculation of A. chroococcum enhanced the plumule length of Chinese cabbage significantly by 1.75 and 2.25 cm under salinity of 50 and 100 mM of NaCl, respectively. Similar results were shown by Kim et al. (2012), they found that co-inoculation of Brevibacterium iodinum and Methylobacterium oryzae mitigated the salinity stress and promoted the root length of maize and sorghum seedling by 22.9% and 29.7%, respectively. However, under salinity level of 150 and 200 mM of NaCl, A. chroococcum did not show positive effect on the plumule length. LAPmix inoculation improved the plumule growth significantly by 1.8 and 1.4 cm at salinity treatment of 50 and 100 mM of NaCl, respectively. Priming with PGPR increase germination and improve seedling establishment. It initiates the physiological process of germination, initiation of physiological process helps in the establishment and proliferation of PGPRon the spermosphere (Taylor and Harman, 1990). Treatments of 150 and 200 mM of NaCl LAPmix inoculation increased the plumule length insignificantly by 0.11 and 0.5, respectively. Lactobacillus sp. inoculation increased the plumule length significantly by 0.7 and 1.2 cm rather than uninoculated seeds at salinity of 50 and 100 mM of NaCl, respectively. However, at salinity stress of 150 and 200 mM of NaCl Lactobacillus sp. inoculation did not show significant increase as well as A. chroococcum and P. putida (Table 3). Tripathi et al. (1998) reported that the phytohormone indole acetic acid production was not affected by salinity as much as nitrogenase production. Actually, low levels of salinity stimulate indole acetic acid production. But at higher than 100 mM NaCl, IAA production was suppressed. Consequently, low level of salinity increases IAA production that may induce roots proliferation and help in the uptake of proline or betaines secreted by the plant roots. Treatment of chickpea seeds with P. fluorescens (Pf1) through seed followed by root zone application after 30 days of sowing increased seedling emergence (Vidhyasekaran and Muthamilan, 1995). More than 33 products of PGPR have been registered for commercial use in greenhouse and field in North America, the threat of certain PGPR (P. aeruginosa, P. cepacia and B. cereus) to infect human beings as opportunistic pathogens has to be clarified before large scale acceptance (Nakkeeran et al., 2005).
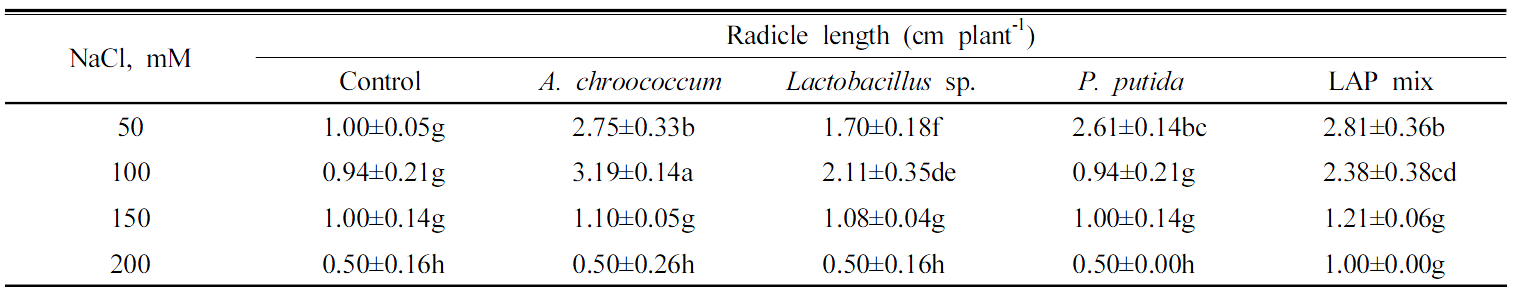
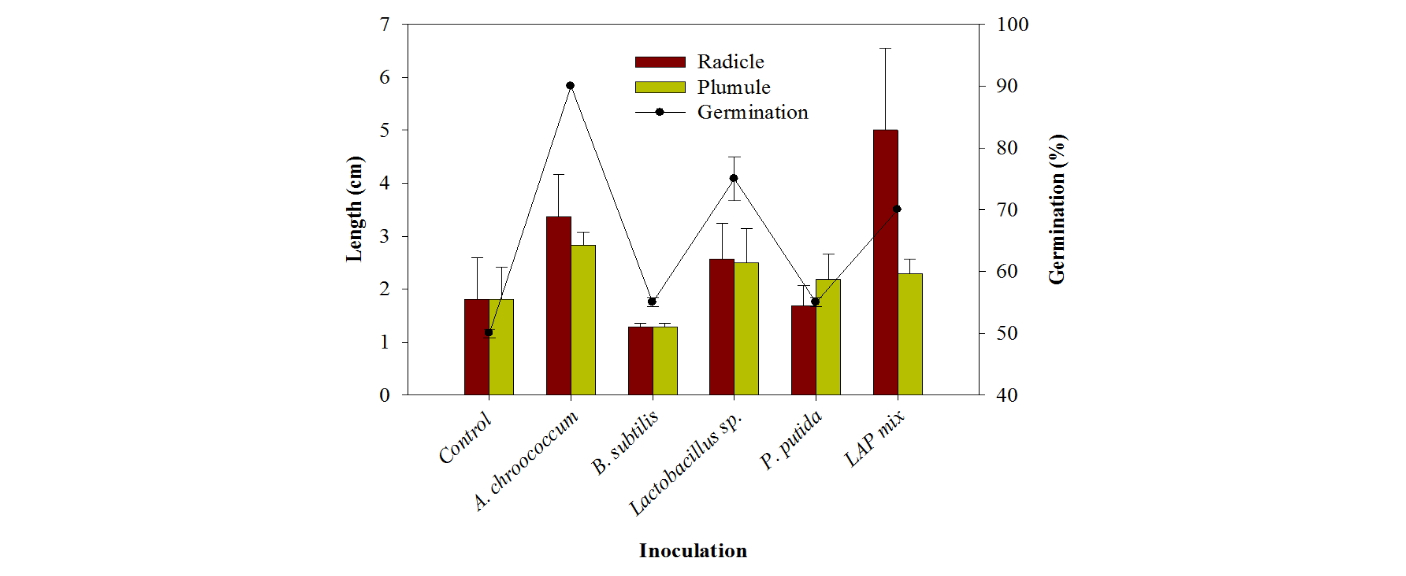
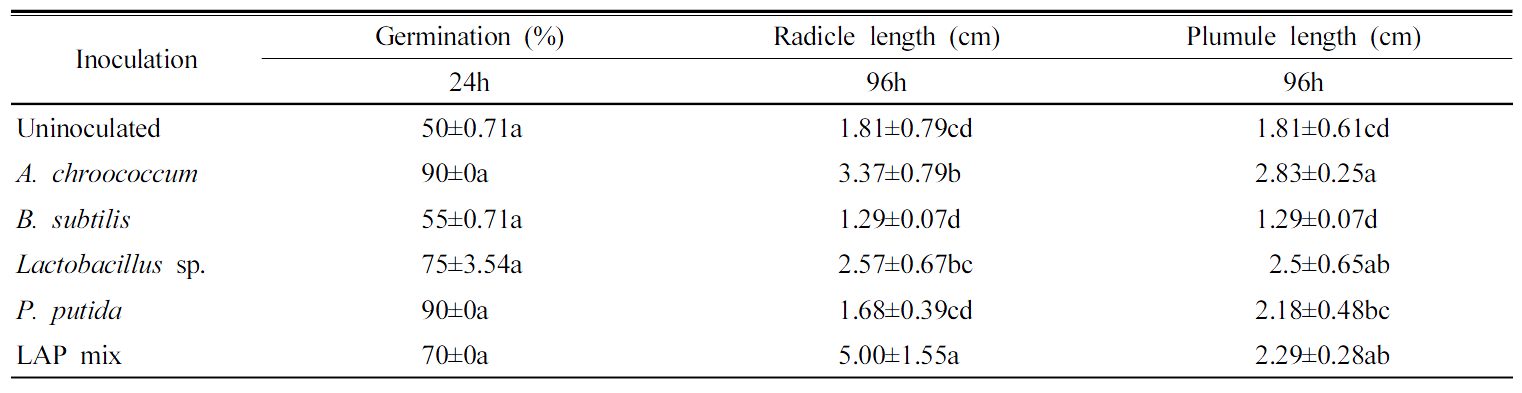
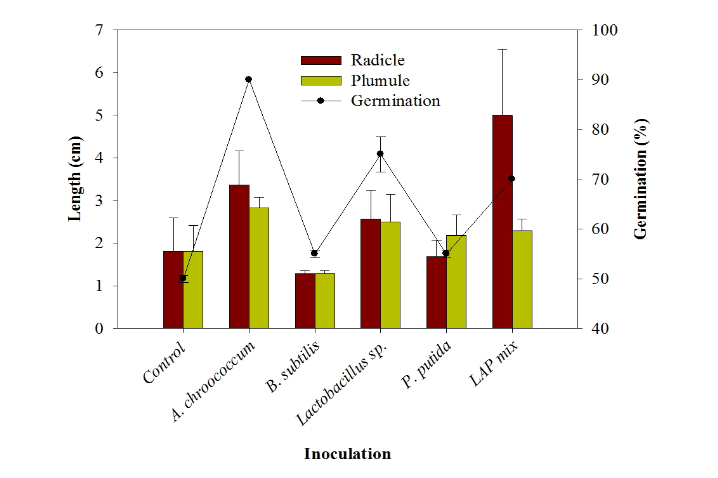
PGP bacteria effect on seeds germination under normal conditions The highest rate of germination of Chinese cabbage seeds after 24 h was 90% by A. chroococcum and P. putida followed by LAPmix which shown 70% of seeds germination after 24 h. However, Lactobacillus sp. showed only 55% (Fig. 2). The uninoculated treatment showed only 50% germination (Table 4). A. chroococcum inoculation enhanced the radicle length by 1.5 cm higher than the control. Lactobacillus sp. enhanced the radicle length by 0.7 cm, while the highest radicle length was shown by LAPmix inoculation which increased the radicle by 3.2 cm higher than the uninoculated control. Surface application of Pseudomonas to beet leaves actively compete for amino acids on the leaf and inhibited spore germination of Botrytis cinerea, Cladosporium herbarum and Phomabetae (Blakeman and Brodie, 1977). A. chroococcum inoculation enhanced the plumule growth by 1.0 cm longer than control. Lactobacillus sp. enhanced the plumule length by 0.7 cm, while P. putida did not show increase either in radicle or plumule length during Chinese cabbage germination. B. subtilis had suppressive effect of the germination process (Table 4). Knowledge on the influence of biotic and abiotic environment on PGPR strains to express its plant growth promotion action has to be studied in badly under in vivo to improve the efficacy of PGPR strains (Nakkeeran et al., 2005). The potentiality of the PGPR strain in the promotion of seed germination should be carried out atboth lab and field conditions in different soil types, microbial communities and climatic conditions (Roberts and Lohrke, 2003). It would lead to the development of applicable PGPR strains (Nakkeeran et al., 2005).
Conclusions
The study presented strains of IAA-producers rhizobacteria (A. chroococcum, Lactobacillus sp., and P. putida) that improve germination rate, root and plumule growth of Chinese cabbage grown under salinity stress. The studied strains have accelerated tolerance of Chinese cabbage under different salinity stresses, and ultimately improved germination, radicle, and plumule growth comparing to uninoculated control. Mostly, the co- inoculation of PGPR showed better results in terms of early germination. The results of this study are encouraging for use of rhizobacterial strains; particularly those possess plant growth promoting traits, to elevate the plant tolerance against salinity. However, further research is still needed to evaluate the PGPR role in the scale soil reclamation.