Introduction
As the high concentrations of nutrients from domestic, livestock and industrial wastewaters enter the stream, river, and lake, the overgrowth of algae in the water environment results in the malodor, growth of pathogenic bacteria, and depletion of dissolved oxygen, as a result of the eutrophication. Consequently, it results in the water pollution and detrimental effects on the aquatic ecosystems (Grady et al., 2011).
Blocking the inflow of nitrogen (N) and phosphorus (P) into the water flowing system is essential to the pre-vention of eutrophication and even after the inflow of N and P, they should be rapidly removed. N and P are major limiting factors causing the eutrophication. However, N can be fixed into NH3 by the nitrogen-fixing bacteria and entered into the water flowing system. Therefore, P should be first removed from the water system to effectively prevent the eutrophication (Correll, 1998). Recently, as the environmental pollution resulting from the eutro-phi-cation has been increased, the biological P removal treatment methods are extensively studied (Oehmen et al., 2007; López-Vázquez et al., 2008; Krishnaswamy et al., 2009), due to the much less secondary environmental pollution. The biological P removal in the wastewater treatment processes was performed by the luxury uptake mechanism in which PAO (Phosphorus Accumulating Organisms, PAOs) (Sedlak, 1991; Wang and Yu, 2000; Zuthi et al., 2013) known as storing the phosphate as polyphosphate in the cell accumulate phosphate beyond the their normal needs by alternating anaerobic to aerobic conditions in the processes (Levin and Sharpiro, 1965; Khosh-manesh et al., 2002). Currently, the Comeau-Wentzel model (Comeau et al., 1986; Wentzel, 1989) and Mino model (Mino et al., 1998) are the most widely accepted in the explanation of P removal mechanism from the biological wastewater treatment processes. The development of new process and the best optimization of process, and the improvement of the process conditions have been performed using the single microorganisms in the biological P treatment process (Barnard, 1973; Wang et al., 2011; Zhang et al., 2013; Lee et al., 2016; Yoo et al., 2016).
However, the wastewater treatment processes through the application of the single pure cultures are limited in removing the water pollutants, since the water pollutants are in most cases existing in the mixtures of heavy metals, organic compounds, and nutrients such as N and P rather than in the forms of single species (Zhang et al., 2013; Yadav et al., 2016). Especially, the heavy metals such as Cd, Cu, Zn, Pb, and Ni are toxic to the living organisms in the aquatic ecosystem and their amounts released into the environment are continuously increased due to their mining and manufacture of industrial products (Jung et al., 2004). The ecotoxicology studies have been extensively performed about the quantitative and qualitative inhibition of heavy metals existing in the form of single heavy metals (Cho et al., 2004). Yilmas (2003) reported that the ecotoxicological study of heavy metals in the environ-ment can be utilized as the basic information in the bioremediation of soil and water contaminated with the heavy metals. However, since most of the heavy metals in the environment may exist in mixture forms rather than in single, they should be considered in their future remediation (An et al., 2004; Kong, 2013). The toxic effects of the mixture of more than two heavy metals on the ecology were evaluated in the recent studies (Liu et al., 2007; Xu et al., 2011). Similarly, the interactive toxic effects of pesticides and organic matters in mixtures on the aquatic ecosystem have been evaluated (Lister et al., 2011; Hernandez et al., 2013). Also, the interactive toxic effects of heavy metals in mixtures were evaluated using plant (Horvat et al., 2007) and fishes (Phillips and Hickey, 2010). Little information is available about interactive inhibition effect of heavy metals on the phosphorus accumulating microorganisms (Kim et al., 2016). Therefore, this study was initiated to quantitatively evaluate the effect of single and binary treat-ments of heavy metals (Cd, Cu, Zn, Pb, Zn) on the growth and P removal efficiency of phosphorus accumulating microorganisms (PAO), Pseudomonas taeanensis.
Materials and Methods
Bacterial culturing Pseudomonas taeanensis (Pseudomonas sp. KCTC 22612) used in this study was provided from Korean Collection for Type Cultures, KCTC). The composition of media for the bacterial culture was as follows: basic medium solution, phosphate solution 1000 mg L-1, 1 M Tris buffer solution (pH 7.0 ± 0.2), trace metal solu-tion (D.W. H2O 100 mL, H3BO3 0.03 g, CuSO4⋅5H2O 0.1 g, ZnSO4⋅5H2O 0.2 g, MnSO4⋅H2O 0.2 g, Na2Mo4⋅2H2O 0.04 g dissolved), glucose solution 200,000 mg L-1 (Zafiri et al., 1999). The volume of liquid media was 50 mL in the 100 mL of Erlenmeyer flask and the amount of liquid inoculum in the experimental media was main-tained and incubated at 2% (v/v). The bacterial culture in the liquid minimal media with the initial P concentration adjusted to 20 mg L-1 was incubated in the shaking incubator at 150 rpm and 30oC during the 24 hours. The liquid bacterial culture was periodically transferred to fresh media using LB media and experimental media for the activation of the culture from the three weeks prior to the beginning of the experiment.
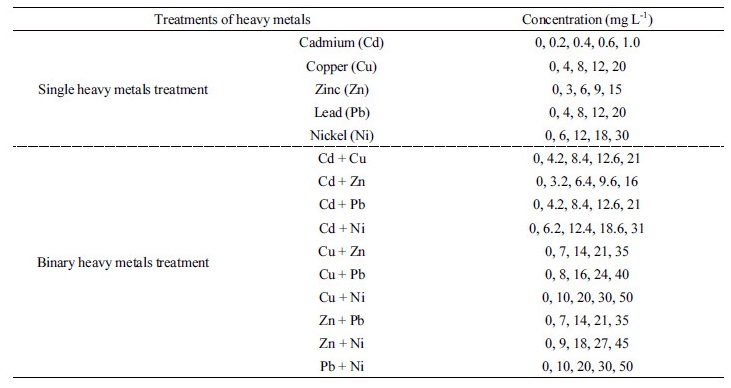
Heavy metal treatment The treatment groups include blank without bacterial culture added in the media, blank with bacterial culture only added, and heavy metals treatments group at the four concentrations of each heavy metal in the form of single and binary mixtures. The standard heavy metals solutions used in the study were Cd standard solu-tion, Cu standard solution, Zn standard solution, Pb standard solution, and Ni standard solution (1,000 mg kg-1, Kanto Chemical Co., Japan). Their concentrations in the treatments were shown in Table 1. The amounts of each one in the binary heavy metals in the experimental media were the same as those in their single treatments. The concen-trations in Table 1 were indicated as the sum of binary heavy metals.
Measurement of bacterial growth and quantification of P Optical density (OD) was used for the measurement of the bacterial growth in the experimental media including bacterial culture. The inoculum for the measurement of OD was sampled using the sterile syringe. The absorbance of the samples taken at once in three hours was measured at the wavelength of 600 nm using UV spectrophotometer (Model UV Mini 1240 Shimadzu, Kyoto, Japan). Also, total P concentrations in the media were calculated from the measurements of their absorbance at the wavelengths of 880 nm using the modification of ascorbic acid-molybdenum blue method (Kim, 2012) and UV spectro-photometer (Model UV Mini 1240 Shimadzu, Kyoto, Japan). The P removal efficiencies in the single and binary treatments of heavy metals were calculated by the measurements of initial and final P concentrations and compared each other.
Estimation of IC50 and EC50IC50 (half of inhibition concentration of bacterial growth) and EC50 (half of effective concentration of phosphorus removal efficiency) was estimated by linear regression equation obtained from the least square of the natural log of absorbance vs time. Log gamma distribution for the estimation of IC50 was determined by fitting data of inhibited growth (Asami et al., 1996). EC50 was estimated at the value that the P removal efficiencies in the heavy metal treatments were half of the control, respectively (Chen et al., 1997).
Evaluation of interactive effects of binary heavy metals treatments As the binary heavy metals were treated in the mixture, the additive toxicity index method (Marking and Mauck, 1975) was used for the evaluation of inte-rac-tive inhibition effects of binary heavy metals. Additive toxicity index method was estimated by the following equation.
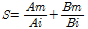
S : sum of toxic response
Am : IC50 of A in mixture of A and B
Bm : IC50 of B in mixture of A and B
Ai : IC50 of A in single
Bi : IC50 of B in single
Based on the S values obtained from the above equation, the interaction of binary heavy metals can be estimated by additive index value (A.I.V.). If S < 1, A.I.V. is 1/S-1.0, if S > 1, A.I.V. is -S+1.0, and if S=1, A.I.V. is S-1 (=0). If A.I.V. > 0, the interaction of heavy metals is synergistic, if A.I.V. < 0, it is antagonistic, and if A.I.V. = 0, it is an additive interaction (Lange and Thomulka, 1997).
Statistical analysis The experimental results were analyzed by SAS software package version 9.1 (SAS Institute, Cary, North Carolina, USA). ANOVA (analysis of variance) and Tukey’s HSD (honest significant difference) test were performed for the multiple comparisons of the results obtained from the treatment groups (Confidence intervals were at 95%). All experiments were performed in triplicates.
Results and Discussion
To evaluate the inhibitory effects of heavy metals treated with single and binary on the growth and P removal efficiencies of Pseudomonas taeanensis, IC50 from the growth inhibition (GI) and EC50 from the P removal efficiencies of Pseudomonas taeanensis were measured and estimated, respectively.
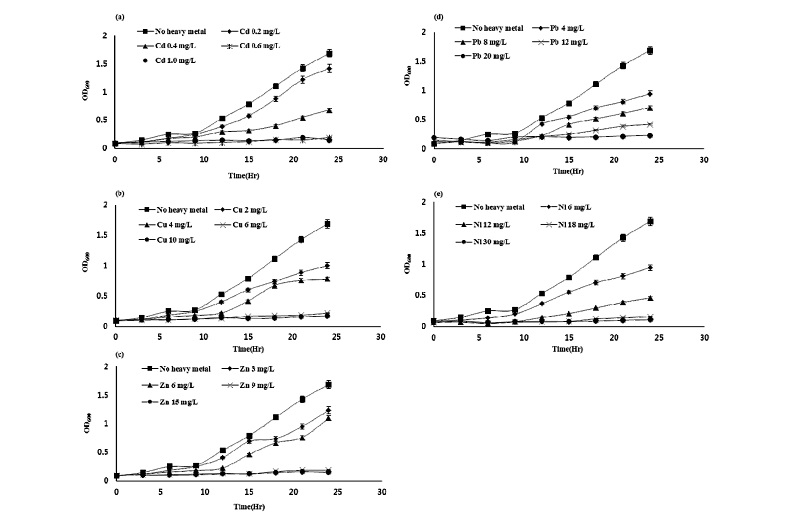
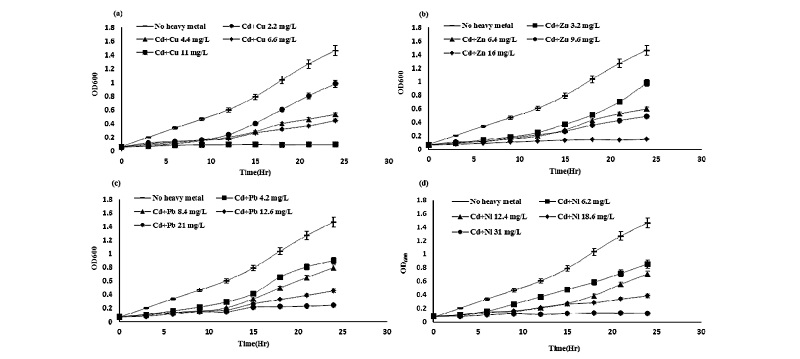
Effect of single treatment of heavy metals on the growth of Pseudomonas taeanensis As shown in Fig. 1, as the concentrations of all heavy metals used in this study were gradually increased, the growth of the bacteria Pseudomonas taeanensis was correspondingly decreased. GI was calculated from the growth curve of bacteria in all treatments of heavy metals. IC50 (mg L-1), which was the concentration of heavy metals corresponding to the half of the growth, was estimated from respective equations obtained from the fitting of the data (absorbance of growth vs concentration of heavy metals) plotted on the growth curve. The inhibitory effect of heavy metals on the growth of Pseudomonas taeanensis was as follows: GI (%) by Cd were 7.1%, 38.9%, 77.1%, and 86.1%, respectively. GI by Cu were 11.6%, 24.9%, 74.2%, and 87.2%; GI by Zn were 9.4%, 46.7%, 74.8%, and 86.8%; GI by Pb were 19.0%, 51.0%, 67.2%, and 93.1%; GI (%) by Ni at wee 3.6%, 31.4%, 78.1%, and 94.7%, respectively. Regardless of the heavy metals used in all the treatments of heavy metals, as the concentrations of heavy metals were gradually increased, the growth of bacteria was correspondingly decreased. IC50 was estimated from GI obtained from the growth curve and their IC50 values corresponding to the treatments of heavy metals were summarized in Table 2. The inhibitory effect of single treatment of heavy metals on the growth of Pseudomonas taeanensis was in the following order: Cd > Cu > Zn > Pb > Ni.
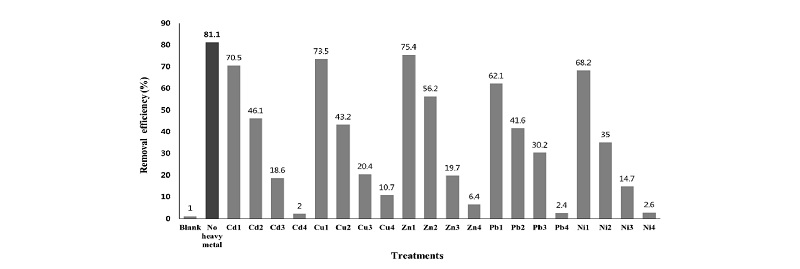
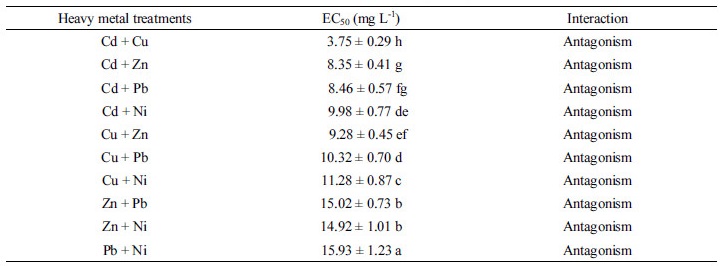
Effect of single treatment of heavy metals on the P removal of Pseudomonas taeanensis The P removal effi-ciencies of Pseudomonas taeanensis were calculated from the initial P concentration and final P concentration and EC50 values were calculated from the P removal efficiencies obtained by the heavy metals treatments and their EC50 values were summarized in Table 3. As in the growth of bacteria, the P removal efficiencies of the bacteria were correspondingly decreased as the concentrations of all the heavy metals used in this study were gradually increased (Fig. 2). Compared to the treatments of heavy metals, the P removal efficiency was the greatest in the control without any heavy metal treatments. The inhibitory effect of heavy metals on the P removal efficiencies of Pseudomonas taeanensis was as follows: Compared to the control, the P removal efficiencies in the treatment of Cd were decreased by 13.1%, 43.2%, 77.1%, and 97.5%; those by Cu were decreased by 9.4%, 46.7%, 74.8%, and 86.8%; those by Zn were decreased by 7.0%, 30.7%, 75.7%, and 92.1%; those by Pb were decreased by 23.4%, 48.7%, 62.8%, and 97.0%; Finally, those by Ni were decreased by 5.9%, 56.8%, 81.9%, and 96.8%, respectively. Based on the P removal efficiencies of the bacteria by the heavy metals applied at the different concentrations, the EC50 of heavy metals were calculated and were summarized in Table 3. The heavy metal the most decreasing the P removal efficiency of the bacteria was Cd, EC50 of which was 0.43 mg L-1, whereas, that the least decreasing the P removal efficiency of the bacteria was Ni, EC50 of which was 11.26 mg L-1. The inhibitory effect of heavy metals on the P removal efficiencies of the bacteria was in order as follows: Cd > Cu > Zn > Pb > Ni.
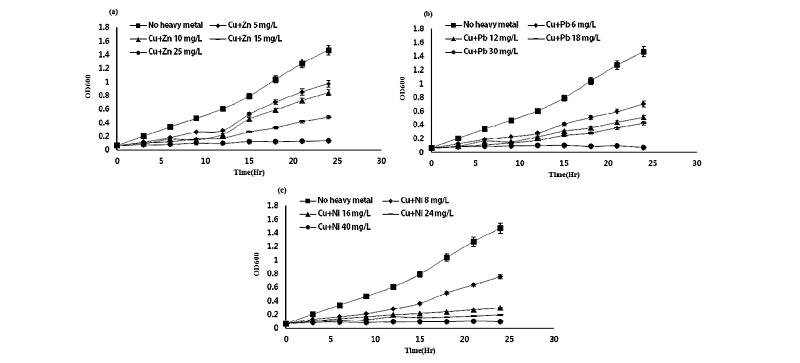
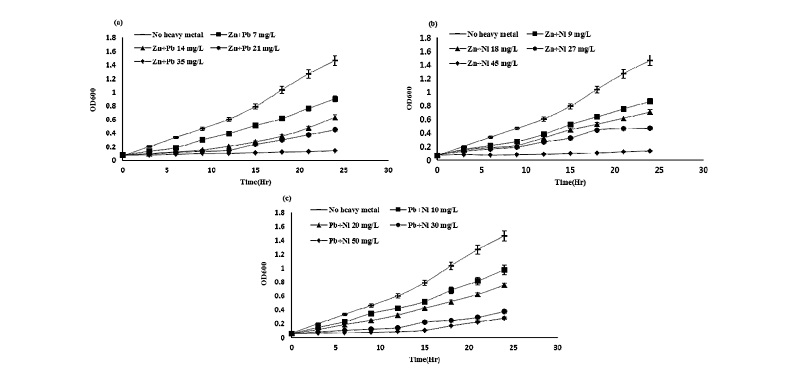
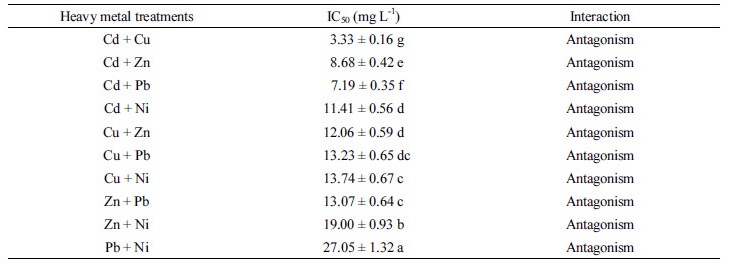
Effect of binary treatment of heavy metals on the growth of Pseudomonas taeanensis To evaluate the inhi-bitory effect of binary mixture treatments of each heavy metals on the growth and P removal efficiencies of Pseu-domonas taeanensis, as the concentrations of heavy metals were applied to the culture, IC50 from the GI (%) of bacteria and EC50 from the P removal efficiencies were estimated, respectively. The interactive effect of the single and binary treatments of heavy metals on the growth of bacteria was evaluated by using the additive index values. On the binary treatments of heavy metals to the bacterial culture, their growth was decreased as the concentrations of binary heavy metals were increased and the time to reach the log phase of the bacterial culture was found to be delayed, compared to the control treatment (Figs. 3-5). Growth inhibition (GI, %) and IC50 values of the bacterial culture by the treatments of heavy metals at different concentrations were calculated from the data fitting of bacterial growth measurement. Also, the single and binary treatments of heavy metals on the bacterial growth were compared to evaluate and compare their interactive inhibition effects on the bacteria, the results of which were shown in Table 4. The binary treatment of respective heavy metals the moat inhibiting the bacterial growth was the Cd+Cu treatment, IC50 of which was 3.33 mg L-1, whereas, that of the respective heavy metals the least inhibiting the culture growth was the Pb+Ni treatment, IC50 of which was 27.05 mg L-1. Based on the evaluation of the inte-ractive effect of the binary treatments of heavy metals on the bacterial growth using the additive index value, compared to these respective single treatments, all the binary treatments of heavy metals were shown to be antagonistic. However, their A.I.D. values ranged from -0.10 in the Cd+Pb treatment to -0.27 in the Zn+Pb treat-ment, whereas, those ranged from -1.02 in the Cu+Zn treatment to -1.08 in the Cu+Pb treatment.
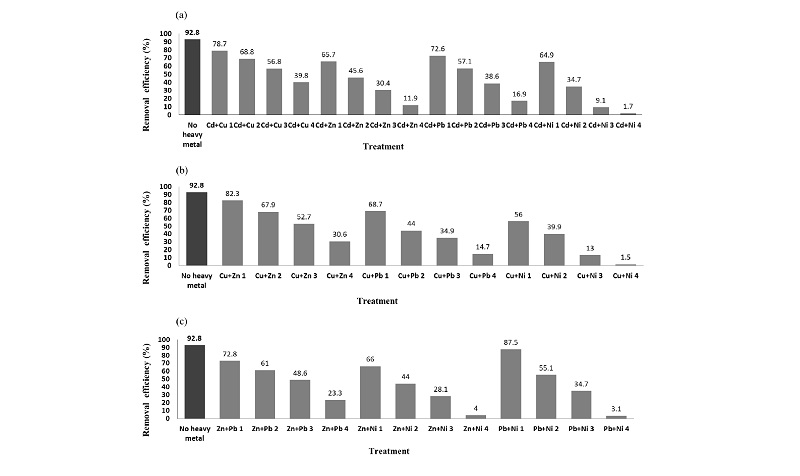
Effect of binary treatment of heavy metals on the P removal of Pseudomonas taeanensis The initial and final P concentrations were measured in the respective heavy metals treatments to evaluate their interactive reduction effects on the P removal efficiencies of the bacteria and the respective P removal efficiencies in all the treatments were shown in Table 5. The P removal efficiencies were correspondingly decreased as the concentrations of heavy metals in all the treatments were gradually increased and they were the highest in the control treatment without heavy metals added (Fig. 6). The P concentrations were measured and P removal efficiencies were calculated and compared in all the treatments. EC50 was estimated by the fitting of the data of P removal efficiencies. Additive index values were used to evaluate the interactive inhibition effect of the binary treatments of the heavy metals on the P removal efficiencies of the bacteria (Table 5). The lowest EC50 was found to be 3.75 mg L-1 in the Cd+Cu mixture treatment, whereas, the highest EC50 was shown to be 15.93 mg L-1 in the Pb+Ni treatment. Based on the evaluation for the interactive inhibition effect of the binary treatments of heavy metals, the antagonistic effects were found in all the binary treatments. Their A.I.D. values were not significantly different.
Conclusions
Pseudomonas taeanensis used in this study for the quantitative inhibitory effects of heavy metals is one of PAO. It was reported in the previous studies that these bacteria can be used in the biological P treatment and showed the high P removal efficiencies under the best optimal conditions (Kim et al., 2010; Yoo et al., 2011; Kim et al., 2012). Pseudomonas taeanensis is generally gram-negative bacteria and do not form the spores and also showed a positive reaction in oxidation and reduction test which is aerobic. The recent studies showed that it can be applied to the actual wastewater treatment for the P removal in the activated sludge process (Cai et al., 2007; Li et al., 2012).
The study was initiated to quantitatively evaluate the inhibitory effect of the five heavy metals (Cd, Cu, Zn, Pb, and Ni) on the growth and P removal efficiencies of Pseudomonas taeanensis cultured in the minimal media. Based on the results from the study, the IC50 and EC50 values were estimated on the single and binary treatments of the heavy metals, respectively. A.I.D. method was used to evaluate the interactions of binary treatments of heavy metals (Kim, 2002).
On the single and binary treatments of heavy metals, as the concentrations of heavy metals were gradually increased, the growth and P removal efficiencies of the bacteria were correspondingly decreased, resulting in the lower IC50 and EC50 values. These results were consistent with those by Diamantino et al. (2001) that the lower IC50 and EC50 were obtained from the study about the inhibitory effect of Zn on the lactate hydrolase activity.
Several publications have focused on the individual effects of heavy metals on the microbial population and activities, e.g., Cu, Zn, Cd, Ag, Pb and Hg (Kim, 2009; Lotti et al., 2012; Yang et al., 2013; Zhang et al., 2015; Kim et al., 2016). Microbial activities may reflect the functions of the total (respiration) or specific (P removal, nitri-fication, and so on) groups of microorganisms in an environment (Domsch et al., 1983). In the experiment eva-luating the inhibitory effects of single treatments of five heavy metals (Cd, Cu, Zn, Pb, and Ni) on the growth and P removal efficiencies of Pseudomonas taeanensis, Cd was found to be the most toxic heavy metal. These results were consistent with those from the study by Kim (2009) that E. coli, archaebacteria, and Pseudomonas aeruginosa was the most sensitive and their growth was the most sensitive to exposure of Cd. Based on the IC50, the growth inhibition of Pseudomonas taeanensis was in order as follows: Cd > Cu > Zn > Pb > Ni. Based on the results from both IC50 and EC50, the resistance to Ni of the single bacterial culture was the strongest of all heavy metals. Similar to the results from this study, Gikas (2008) reported that Ni in the lower concentration was inhibitory to the bacterial growth in the activated sludge process.
Few studies on the joint effect of heavy metals on bacteria have found in the literature (Kimura and Isaka, 2014; Zhang et al., 2015; Kim et al., 2016; Zhang et al., 2016). At low concentrations, both Cu (II) and Zn (II) are neces-sary micronutrients and components of many enzymes and co-enzymes of microorganisms. However, excessive Cu (II) and Zn (II) inhibit or may be toxic to microorganisms in biological wastewater treatment systems (Ochoa- Herrera et al., 2011; Zhang et al., 2015). In this study, it was found that the bacterial growth slowed down and stayed at the longer lag phase in the incubation of bacteria along with binary treatment of heavy metals. IC50 of Pseudomonas taeanensis was 3.33 mg L-1, the lowest in the Cd + Cu treatment and 27.05 mg L-1, the highest in the Pb+Ni treatment, respectively. The results of the present study correspond well with those found in the earlier experimental studies in Alcaligenes sp. (Kim et al., 2016). They have reported that the treatments of lowest IC50 and EC50 were the Cd+Cu treatment in the binary mixture treatments of heavy metals. The results for the interaction of binary treatments of heavy metals based on the EC50 were a little different from those on IC50. The interactive results on IC50 were all antagonistic, whereas, those on EC50 were mostly antagonistic. The results from this study were different from those by Buhl and Hamilton (1997) and Tao et al. (1999) that the mixture treatments of heavy metals showed the additive or synergistic effects in most cases. However, Kim (2002) reported that the toxic effects and patterns of heavy metals mixtures can be different by the use of bioassay methods applied. The results obtained from this study were in most combinations of mixture antagonistic which may be due to the selection and use of PAOs rather than the popular microorganisms such as E. coli and Vibrio fischeri. It was found from this study that antagonistic effects were found in all the binary treatments of heavy metals including Cd. These results were similar to those that the binary mixture of heavy metals including Cd can decrease the toxicity of cell in the toxicity test (Shopsis, 1994; Sharma et al., 1999).
Based on the results obtained from this study, it appears that they could provide the basic information about the toxic effects of the respective individual and binary treatments of heavy metals on the growth and P removal effi-ciency of other PAOs through a further study about the characterization of functional proteins involved in toxic effects of heavy metals.