Introduction
Materials and Methods
Composting system and materials
Gases sampling and analysis
Compost sampling and chemical analysis
Statistical analysis
Results and Discussion
Changes of temperature and pH
Gas emissions & Global warming potential (GWP)
Nutrient contents and maturity
Conclusions
Introduction
With the rapid development of the population and economy, huge amounts of food waste have been generated. Recent report shows that 5.8 million tons per year (2017) of food waste is generated in Korea (MOE, 2019) and is expected to rise more in the next two decades. The food waste disposal has become increasingly serious and needs to be solved urgently. The improper treatment will cause substantial production of leachate and greenhouse gases, damage biodiversity and affect the living environment of residents. Since 2005, however, landfilling of food waste (FW) was prohibited in South Korea, most FW (86% of total production amount) is recycled by composting and converted into feed products and fertilizers (MOE, 2019). Aerobic composting is regarded as one of the most economically efficient pathways to treat and recycle food wastes (Chan et al., 2016; He et al., 2018).
Composting treatment is a well-developed technology adopted by various countries. By composting process, the organic waste is transformed into a stable organic matter. However, the efficiency of FW composting is often very low due to its low porosity, high bulk density, poor C:N ratio, easy acidification, and others (Awasthi et al., 2017). These limitations could be overcome by combining it with other organic wastes like livestock manure, sludge, and others.
The vast amounts of livestock manure is another issue caused by the intensive development of livestock industry. According to a recent report, 153 thousand tons livestock manures per day is produced in Korea. The amount of manure (thousand ton per day) generated by livestock type was cattle (63) > swine (61) > poultry (28) in that order. The livestock manure (LM) has been mostly treated by aerobic composting and anaerobic digestion, to produce biofertilizer and bioenergy for resource recovery (Luo et al., 2013; Liu et al., 2020).
The combining of food waste and manures, which are continuously increasing, not only reduces the time and labor but also saves the costs, compared to separated disposal of each waste. A few studies reported combination effects of livestock manure and food waste, such as swine manure, chicken manure (Hwang et al., 2020b; Ravindran et al., 2022), and most of food waste composting research focused on bulking agents like rice husk, biochar, vermiculite, mature compost (Chang and Chen, 2010; Chen et al., 2010; Cao et al., 2018; He et al., 2018; Yang et al., 2019). However, the comparison studies among manures types have not been fully conducted, and detailed impact of the different livestock manure types on gas emission during composting process is poorly understood.
Therefore, the current research was designed to investigate the influence of co-composting of various livestock manures and food waste. The changes in temperature, pH, maturity, chemical properties of compost piles were monitored, and GHGs (CH4 and N2O), NH3 emission were analyzed to develop practical food waste composting strategy. The results of this work reveal an effective practice for alleviating gas emissions and improve compost quality.
Materials and Methods
Composting system and materials
Four of 62 L PVC containers (length × width × height = 650 × 440 × 510 mm) covered with thick polystyrene were installed to prevent heat loss. The four treatments were not replicated since the composting scale (62 L of volume) ensures the experimental reproducibility as well evidenced in other studies (Yuan et al., 2015; Jiang et al., 2016; Zhang et al., 2016; Hwang et al., 2020a). Air was supplied from the bottom of the container chamber with a flow meter (1 - 1.5 L min-1). The four treatments were labeled as OF (only food waste (FW), control), FCM (FW + cow manure), FSM (FW + swine manure) and FPM (FWM + poultry manure), respectively. The FW and manures were collected from a local municipal waste station, and farm houses, respectively. The characteristics of the raw materials used for this experiment are described in Table 1. Raw and fresh materials were manually mixed with the ratio of 35% food waste and 35% livestock manure, and 30% sawdust (dried w/w) was used to control initial moisture content and C/N ratio. Moisture contents of raw materials were determined before the test to make same dried weight of compost pile (25 kg), and control water content between 60 and 65%, because the water content of compost pile is very important for composting process. Since there is little difference in moisture content among manures, the final moisture contents of compost pile were recorded around optimum range. For OF treatment, 70% of food waste and 30% of saw dust was mixed. Temperature sensors was placed in middle of compost pile and, the internal temperature was monitored every 12 h using a data logger (EM50 Data logger, USA).
Table 1.
Characteristics of the composting materials used for this experiment.
Gases sampling and analysis
The closed chamber method was used to determine GHGs such as methane (CH4) and nitrous oxide (N2O) at 1, 8, 15, 22, 29, 36, 43, 57, 71 and 85 days during composting period (Jeong et al., 2018; Hwang et al., 2022). The concentrations of two gases were measured using gas chromatography (Shimadzu, GC-2010, Tokyo, Japan). CH4 and N2O concentrations were determined by a flame ionization detector (FID) with a methanizer and a 63Ni electron capture detector (ECD), respectively.
The gas was extracted by air pump from composting bin for six hours. The uptake of gas-phase NH3 by sulfuric acid was measured by aqueous concentration of ammonium in the sulfuric acid. The increased NH3 uptake in acid solution is apparently due to reaction between NH3 and H+ at the gas-liquid interface. Concentration of ammonium sulfate was analyzed by auto analyzer 3 (Bran Luebbe, Germany).
Gas emission were calculated from gas concentration per compost weight over a specific time interval. Total fluxes of gases were calculated on a mass basis during composting process (g kg-1). Details related to the sampling and calculation of gases are explained in a previous study (Han et al., 2018; Hwang et al., 2020b).
Compost sampling and chemical analysis
The compost was manually turned over and properly mixed prior to collection on 1, 8, 15, 22, 29, 36, 43, 57, 71, and 85th day to homogenize the compost pile. After mixing, compost samples were collected using a core, and divided into two parts; one portion was preserved at 4°C before analysis, and the other portion was air-dried at 65°C for approximately 48 h, ground and sieved with a 2 mm mesh for chemical analysis.
The pH and electrical conductivity (EC) values were determined in the compost:distilled water suspension (1:20, w/v ratio). The extract was filtered through a 0.45 µm membrane filter to analyze the dissolved carbon and nitrogen concentration; Concentration was determined using a TOC-5050A analyzer (Shimadzu Corporation, Tokyo, Japan). The total carbon and nitrogen contents were analyzed by element analyzer (CHNS-932 Analyzer, Leco). Fresh samples were extracted with distilled water (1:10), and filtered through a 5 µm filter paper to evaluate the germination index (GI). GI value was used to assess the phytotoxicity and maturity level of compost (Yang et al., 2015; Chen et al., 2019). The GI was measured and calculated according to RDA (2012). A segmented flow analyzer (Technicon Autoanalyzer II System, Germany) was used for quantifying NH4+-N and NO3--N in composting materials extracted by mixing fresh samples with 2 M KCI solution at a ratio of 1:10 (weight/volume).
Statistical analysis
Mean values and standard deviations of triplicate measurements were shown in this study. The data were subjected to one-way analysis of variance (ANOVA), and identified the least significance variance (LSD) at P = 0.05 values by Statistical Analysis System (SAS 8.2).
Results and Discussion
Changes of temperature and pH
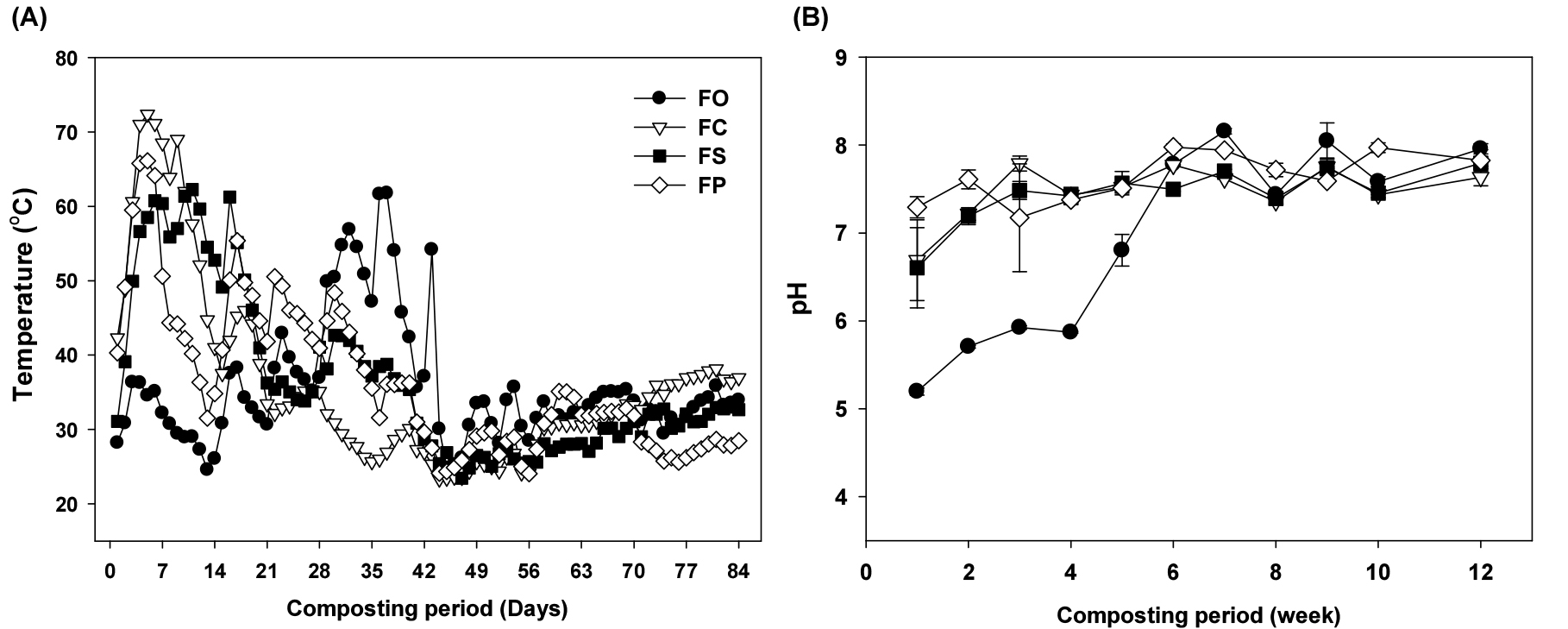
Temperature was considered as the essential parameter because it could reflect the organic matter degradation and microbial activity of composting process. The temperature changes of compost pile showed three stages, namely thermophilic (>55°C), mesophilic stage and maturation stage, which is generally divided in composting experiment (Jäckel et al., 2005; Mehta et al., 2014) (Fig. 1A). The changes in temperature of manure combination treatments displayed similar tendencies. After the test started, it increased rapidly, reaching the thermophilic phase within 4th day. The highest peak was 72, 61, 66°C in FCM (5th), FSM (6th) and FPM (5th), respectively. The OF treatment showed the peak 57°C on 32th day, and the period of thermophilic was relatively shorter (for 3 days) than FSM (10 days), FCM (9 days), and FPM (4 days). It can be attributed to that the addition of LM could enhance microbial activity and then accelerate the decomposition of composting materials (Chen et al., 2017). Afterwards all treatments showed a gradual decline, and control (OF) mostly recorded the lowest temperature the composting period.
The pH of compost is also an important factor that affects microbial activity during composting. Except OF, similar patterns of pH values were observed in FW + LM treatments. It rose within 15th days then it maintained between 7 to 8.5, which is commonly considered as an optimal pH value for efficient composting (Wong et al., 2009) (Fig. 1B). The initial pH value of OF was very low (5.2), and it reached pH 7 at 6th week, middle of composting process. The pH of all treatments slightly decreased at the beginning stage possibly due to the production of organic acids, which agrees with the results reported by Yang et al. (2019). Increasing trend in the thermophilic phase is due to the release of organic and inorganic acids during the degradation of organic materials (Li et al., 2020; Wang et al., 2021). At final stage, all treatments showed relatively stable and higher than initial value. The pH value of OF increased from 5.2 to 8.0, FCM: 6.7 to 7.6, FSM: 6.6 to 7.8 and FPM: 7.3 to 7.8, respectively.
Gas emissions & Global warming potential (GWP)
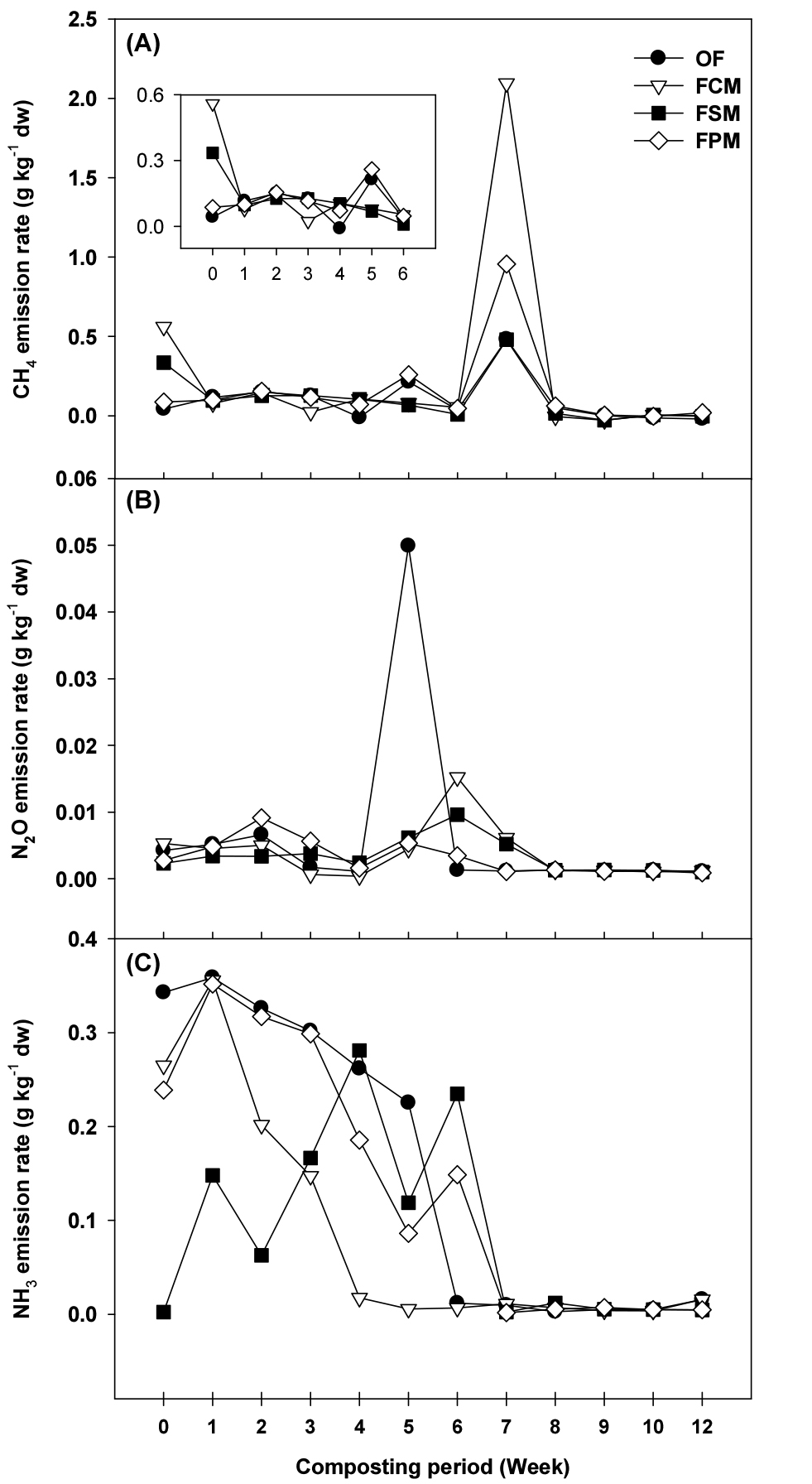
Although aerobic composting emitted less CH4, it is main GHG generated during composting due to the 25 times higher global warming potential (GWP) than that of CO2. The CH4 emission patterns were similar among treatments. After the experiment started, the CH4 flux of FCM and FSM was rapidly produced, but immediately decreased like other treatments (Fig. 2A). The initially increased CH4 emission might be due to the largely consumed oxygen for organic matter decomposition during the thermophilic beginning phase. The highest peak was observed at 7th week in all treatments; FCM (2.10 g kg-1 dw), FPM (0.96 g kg-1 dw), OF and FSM (0.48 g kg-1 dw). The emission patterns indicated that the anaerobic condition caused similar CH4 emission pattern, but concentration was different among the treatments. Methane was produced by methanogen using CO2 and acetic acid in anaerobic condition. Higher emission of CH4 in FCM and FPM might be indicated the unsuitable aeration during composting and improper density between raw materials.
The changes in N2O emission are shown in Fig. 2B. The N2O production occurs by nitrification and denitrification of compost materials (Shen et al., 2011; Wu et al., 2017). Overall, low mean N2O emissions were recorded during whole composting period. High emission was once observed in OF and slightly high peak in others at middle of composting process, while tended to the baseline in other days (Fig. 2B). Wang et al. (2018a) reported that relatively lower temperature and pH of compost was favorable condition for N2O emission during thermo and mesophilic phases, which agreed with N2O emission pattern in this study.
The curves of NH3 content are shown in Fig. 2C. The NH3 mainly occurred within first 30 days, which accounted for 62 - 81% of total flux. It is due to the volatilization of ammonium ions present in the initial compost pile under high temperature condition. And with the rise in temperature and pH value of the compost material, the rapid decomposition of organic matter resulted in the release of large amounts of NH3 in initial composting phase (Figs. 1 and 2). This emission pattern agreed to previous studies (Zhang et al., 2016; Wang et al., 2018b; Yang et al., 2019; Hwang et al., 2022). The changes in NH3 were strongly correlated with NH4-N and DOC (Fig. 3). It might be attributed to the conversion of NH4+ to NH3, which was caused by the rapid degradation of organic N to inorganic N. Ammonium would subsequently volatilize under high temperature and pH conditions.
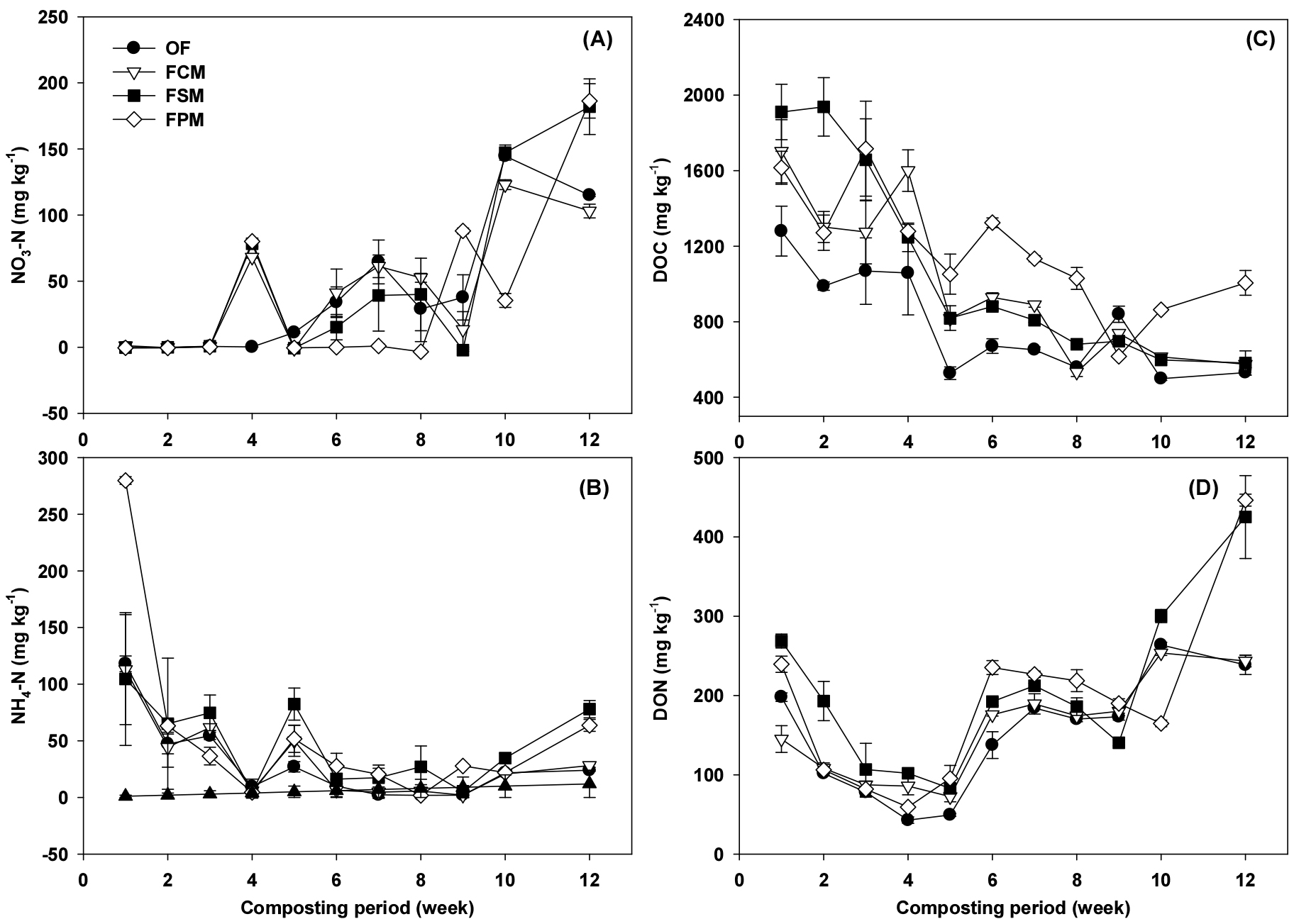
Fig. 3.
Changes in different chemical parameters during composting process: (A) Nitrate nitrogen, (B) Ammonium nitrogen, (C) Dissolved organic carbon (DOC) and (D) Dissolved organic nitrogen (DON). FO, food waste only; FC, food waste + cow manure; FS, food waste + swine manure; FP, food waste + poultry manure. All data are the mean of three replicates, and eror bars indicate standard deviations.
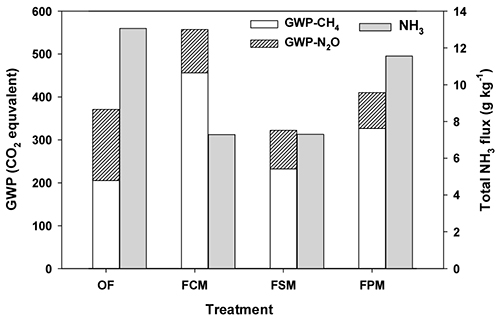
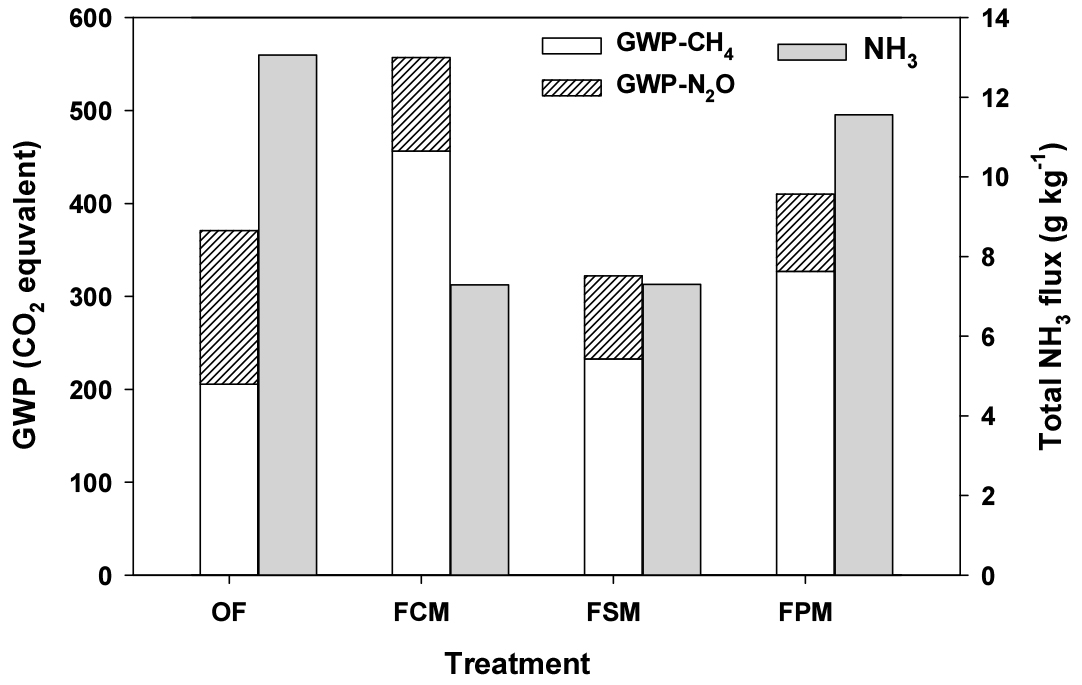
Fig. 4 showed global warming impact and NH3 fluxes of all treatments. The total GWP were expressed as CO2-C equivalents using a GWP values of 1 for CO2, 25 for CH4, and 298 for N2O (IPCC, 2007). The total GWPs of FCM, FPM, OF and FSM treatment were 14.7, 9.2, 7.6 and 7.0 kg CO2 eq. kg-1, respectively. In manure treatments, the CH4 was main contributor on total GWP ranged from 79 to 89%, but GWP of FO was composed with 65% CH4 and 35% N2O emission. The LM combination slightly increased CH4, however, effectively reduced N2O (39 - 50%) and NH3 losses (12 - 44%) during FW composting process, compared to OF. These results showed that swine manure was the most recommendable material to control GHGs and NH3 emissions for food waste composting.
Nutrient contents and maturity
The changes in dissolved carbon and nitrogen (DOC and DON), and the NH4+-N and NO3--N throughout the composting process are shown in Fig. 3. Low NO3--N concentrations were observed in the thermophilic phase in all treatments (Fig. 3A) since the nitrification process was hindered by high NH3 toxicity and temperature (Paśmionka et al., 2021). The activity of nitrifying bacteria could be promoted under 40°C and aerobic condition (Zhang and Sun, 2015; Awasthi et al., 2020). Therefore, nitrification did not occur, and resulted in low NO3--N content during thermophilic stage (Fig. 3A), then it increased from 35th day, when NH3 emission started to decrease. The NH4+-N decreasing trend might be due to the ammonia volatilization and the conversion from NH4+ to NO3- (Fig. 3B), because NH4+ ion was used for both NH3 volatilization and nitrification. Since there was a lot of NH3 volatilization in the early phase, nitrification action would not have occurred due to low content of NH4+. Ammonia volatilization is a chemical reaction, and nitrification is a biological reaction involving microorganisms, volatilization action could occur first.
The dissolved organic carbon (DOC) concentration was higher during initial stage, and decreased from 4th week because microorganisms greatly decomposed the available carbon under thermophilic phase (Wang et al., 2013). The DOC concentration has positive correlation with temperature (Fig. 3C). The dissolved organic nitrogen (DON) contents were fluctuated during whole composting period, but trends in DON changes were similar for all treatments (Fig. 3D). The C/N ratio increased at final stage due to nitrogen loss during composting process, and the highest C/N ratio was obtained in the FO (Table 2), which emitted the largest amount of NH3. The increase in nutrient content such as Ca, Mg, K, Na may also be attributable to the so-called ‘‘concentration effect’’ in which the organic matter was lost in the form of gases such as H2O and CO2, resulting in an increase in the nutrient content per unit mass (Table 2) (Chi et al., 2020).
Table 2.
Characteristics of initial and final compost piles.
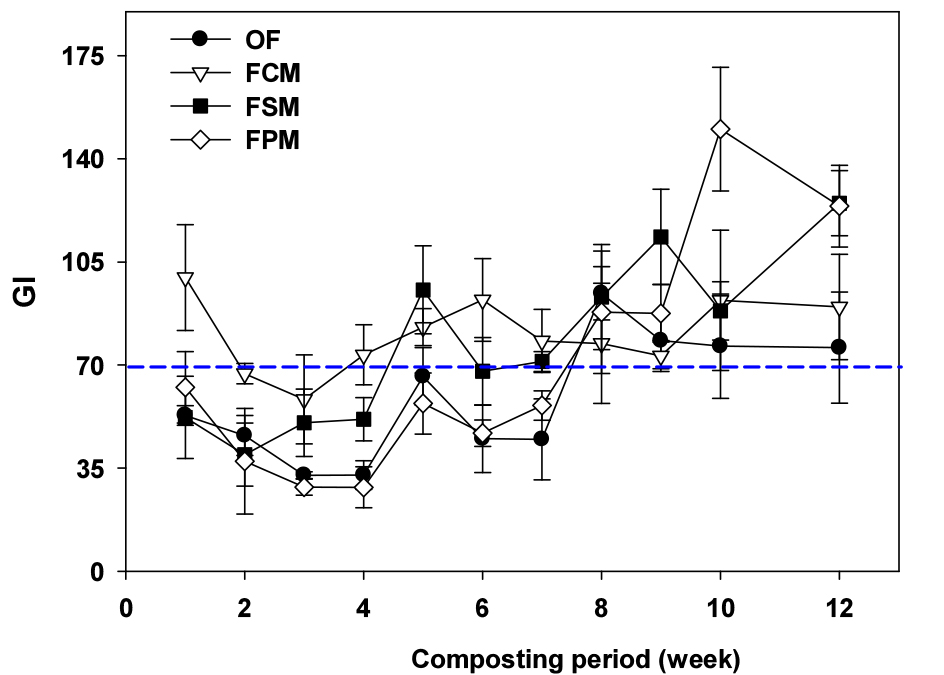
The maturity and phytotoxicity degree of compost were determined by germination index (GI) value. In all treatments, the GI values gradually increased with composting process (Fig. 5). The GI value of FO and FC was fluctuated between 7th and 10th weeks, but FC and FP treatments reached above 80%, indicating maturity (Zhang et al., 2018; Duan et al., 2021), at 8th week. This result showed FW co-composting with swine and poultry manure can shorten composting period.
Conclusions
Combinations of different livestock manures and food waste were investigated to understand their co-composting synergistic effects on GHGs and NH3 reduction and compost quality during composting period. This research showed that mixing manure promoted composting process by prolonging the thermophilic phase, increasing temperature, and reduced the NH3 during kitchen waste composting. The co-composing with swine manure was the most effective to reduce NH3 loss. In further research, absorption mechanisms of swine and cow manures and microbial interaction between manures and FW still need to be studied.