Introduction
Kiwifruit (Actinidia spp.) is a deciduous vine fruit tree cultivated in New Zealand, Italy, China and Korea (Koh et al., 2005; Luongo et al., 2011). Kiwifruit is classified as A. deliciosa (Green-fleshed kiwifruit) and A. chinensis (Yellow-fleshed kiwifruit), depending on flesh color and the existence of hair (Montefiori et al., 2009). Green- fleshed kiwifruit, Hayward has been widely cultivated worldwide and the global quantity of kiwifruit cultivation has greatly increased since the late 1980s (Crisosto et al., 2012). In South Korea, kiwifruit has been cultivated since the early 1980s and the enormous quantities of it have been marketed and exported successfully, due to medicinally useful antioxidants such as vitamin C, carotenoids, flavonoids, minerals and others (Wills et al., 1986). Production quantities of kiwifruit also rapidly increased because of vitamin C contents in kiwifruit that captured the public’s imagination. However, kiwifruit has experienced damage by frost and severe cold in November which is the month of harvest, and after that time, postharvest diseases originating from fruit rot fungi infection are serious problems (Koh et al., 2002; Shim and Ha, 1999). On average, the incidence of postharvest diseases in percentage terms was 32%, while internal and external symptoms were 21.9% and 4.9%, respectively. Additionally, both symptoms affected 5.2% of the kiwifruit crop in South Korea (Koh et al., 2005). The kiwifruit output of other countries is much superior to that of South Korea, and therefore the international competitiveness of the kiwifruit industry is important (Cho et al., 2011). Accordingly, new cultivars have been developed to supplant any weaknesses in existing cultivars and ensure that exports outweigh imports (Park, 2009). For example, green-fleshed kiwifruit cv. Gamrok has higher sugar content and its harvest time has been brought forward from mid-November to late October. Yellow-fleshed kiwifruit cv. Goldone does not require regulations on enlarging the fruit size (Kwack et al., 2012). Red-colored and yellow-fleshed, cv. Redvita has higher vitamin C content than existing cultivars (Kwack et al., 2016). Both cvs. Goldone and Redvita have also had their harvest periods brought forward from mid-November to October.
Postharvest fruit rot diseases accelerate as ethylene is secreted during ripening, the sugar content is higher and latent pathogens generate postharvest disease (Koh et al., 2005). For example, in New Zealand, the three-major postharvest fruit rot diseases of A. deliciosa are: ‘Field rot’ caused by Sclerotinia sclerotiorum; ‘Storage rot’ caused by Botrytis cinerea; and ‘Ripe rot’ which is caused by Botryosphaeria dothidea (Pennycook, 1985). In the United States, postharvest diseases observed in kiwifruit are ‘Botrytis rot’ caused by B. cinerea, ‘Alternaria rot’ due to A. alternata, ‘Phomopsis stem-end rot’ caused by Diaporthe Actinidia and ‘Blue mold’ which is blamed on Penicillium expansum (Sommer et al., 1983). Postharvest fruit rot diseases reported in South Korea are ‘Ripe rot’, ‘Stem-end rot’ and ‘Gray mold’; their respective generated ratios of infection are 83.3%, 11.9% and 1.4%, respectively (Koh et al., 2005). Recently, ‘Sclerotinia rot’ caused by S. sclerotiorum (Lee et al., 2015), and ‘Black rot’ caused by Nigrosporea sp. (Kwon et al., 2017) were reported as postharvest diseases of Kiwifruit.
Symptoms of postharvest diseases mostly appear as internal symptoms but external symptoms sometimes appear as soft skin and surface covered by gray or white mycelium. Even if external symptoms are not evident, decay as an internal symptom appears as green lesions caused by water immersion when the skin is peeled off (Koh et al., 2005). Symptoms of postharvest diseases reported in Korea seem to differ in terms of fungi. Symptoms of ‘Ripe rot’ form shallow and brown dimples on the sides of the fruits and these dimples rapidly expand to large oval lesions as fruits ripen. Usually, there is only such lesion is but sometimes there can be up to three per fruit. Symptoms of ‘Stem-end rot’ are similar to ‘Ripe rot’ in the way that water-soaked lesions exist but white mycelial mats and exudates are formed on the stem-ends of fruits. Fruits affected by ‘Gray mold’ decay while in storage or when they are being sold. The fruit skin is covered by gray mycelial mats, and fruit flesh is very soft and in severe cases may be in a liquefied condition. Lastly, ‘Sclerotinia rot’ appears as soft and water-soaked lesions on fruit flesh but the fruit skin is covered with white mycelial mats.
Hawthorne et al. (1982) reported that the ripening period affects rates of postharvest disease. On two separate occasions, kiwifruits were stored at 0~1°C for one month and ripened at 15~20°C for 2 and 5 weeks, respectively. When kiwifruits were ripened for 2 weeks, detection ratio of pathogens from postharvest diseases was 32%, but when kiwifruits were ripened for 5 weeks the ratio was 75%. Despite the three-week interval, diseases rates showed obvious differences. Watanabe and Takahashi (1998) surveyed the firmness and brix degree of kiwifruits in cultivars. The yellow-fleshed cv. Golden King and cv. Yellow Queen samples grew faster and accumulated brix faster than the green-fleshed cv. Hayward and Bruno varieties. Flesh firmness of the yellow-fleshed cultivars declined faster than that of the green-fleshed cultivars. The fruits continued to soften in cold storage, and the rate was also faster in the yellow-fleshed cultivars than the green-fleshed cultivars. Therefore, storage and ripening conditions were considered as important factors in the extent of diseases in kiwifruit.
Many studies on the diseases of existing cultivars have been done, yet analyses of new cultivars are few. Thus, we verified resistance levels of new cultivars to postharvest diseases on two separate occasions. In the first experiment, cvs. Hayward, Gamrok, Golone and Redvita were stored at 0~1°C for one month and ripened at 16°C for two weeks. The new cultivars were compared to the existing cultivar cv. Hayward. In the second experiment, Hayward and new cultivar Gamrok were stored at 0~1°C for six months and checked for postharvest fruit rot with a month interval. The results provided latest information on protecting commercial kiwifruit cultivars.
Materials and Methods
Sample collection New Cultivars Gamrok, Redvita, and Goldone were developed by the National Institute of Horticultural and Herbal Science (NIHHS) in 2013, 2012 and 2011, respectively. In the first experiment, four different cultivars of kiwifruits were harvested in an orchard located in Sacheon, Republic of Korea. The cultivars Hayward, Gamrok, Redvita and Goldone were harvested on November 13th 2014, November 5th 2014, October 26th 2014 and October 24th 2014, respectively. Fruits were cultivated unscratched and healthy fruits in the same condition were stored. The fruit samples for postharvest disease evaluation were stored until December 2nd 2014 at 0~1°C and ripened for 14 days at 16°C. In the second experiment, kiwifruits, cv. Hayward and cv. Gamrok, from the same orchard were harvested and forty fruits were stored on November 4th 2015. For each cultivar, forty fruits were diagnosed that same day, the remainder were stored at 0~1°C and checked for postharvest fruit rot with a month interval until April 4th 2016.
Disease diagnosis, isolation of fungi The method used was based on that employed by Kader (2002). To diagnose postharvest fruit rot diseases, we observed external symptoms first and peeled of all the skin to identify internal symptoms. Internal symptoms were identified as having soft skin and water-soaked lesions. Diseased parts were separated into 0.5 × 0.5 cm rectangular shapes and washed in 70% ethanol for 1 min, 1% NaOCl for about thirty seconds and washed five times with distilled water. Disinfected parts were eliminated moist on filter paper and pure cultured on Water Agar (WA, Agar 20g per 1L) medium at 27°C for three days. The fungal hypha was transferred onto Potato Dextrose Agar (PDA, Potato extract 4 g, Dextrose 20 g, Agar 15g per 1L) medium at 27°C for one week as pure culture.
Fungal DNA extraction and molecular identification The DNA of fungi was extracted using the modified CTAB DNA extraction method (Porebski et al., 1997). Experimental procedures were performed as follows. Mycelia cultured on PDA medium were collected in a 1.5 ml E-tube. 500 µL of CTAB buffer (1M Tris HCl [pH 8.0], 5M NaCl, 0.5M EDTA, Hexadecyltrimethyl ammonium bromide) and 4 µL of protenase K were added and mixed using a vortex thoroughly. The homogenates were transferred to a 65°C bath for thirty minutes and the hyphae were mashed every ten minutes. 500 µL of PCI (Phenol:Chloroform:Isoamyl-alcohol = 25:24:1) was mixed with the homogenates and the homogenates were centrifuged at 13,000 rpm at 4°C for 10 minutes. Supernatant liquid containing the DNA was transferred to a new E-tube and the same amount of isopropyl-alcohol was added to the new homogenates. The homogenates were inverted and centrifuged at 13,500 rpm at 4°C for 5 minutes. The supernatant liquid was decanted completely without the pellets and subsequently washed with 500 µL of 70% HPLC ethyl-alcohol. Using the same procedure, residual ethyl-alcohol was removed and dried completely. Finally, the DNA samples were dissolved in 20 µL of TE buffer (1 M Tris-HCl pH 8.0, 0.5 M EDTA) and stored at -20°C.
The polymerase chain reaction (PCR) was conducted in 20 µL of total volume. Reaction mixtures contained 13.8 µL of distilled water, 2 µL of 10x reaction buffer, 200 mM of dNTP, 100 pmol of each forward and reverse primer, 50 ng of template DNA and 0.5 unit of Taq polymerase. The forward primer, ITS-1 (5’-TCCGTAGGTGAACC TGCGG-3’) and the reverse primer, ITS-4 (5’-GCTGCGTTCTTCATCGATGC-3’) were used for amplification (White et al., 1990). The polymerase chain reaction was carried out in a gradient thermal cycler (Bibby Scientific Ltd, Staffordshire, U.K.) controlled to execute the denaturation step of 95°C for 2 min. This was followed by 30 cycles lasting 30 sec at 95°C for denaturation, 30 sec at 50°C for annealing and 1 min at 72°C for extension. The final extension was performed at 72°C for 10 min. At 2 µL PCR products were mixed with 10x loading buffer (Takara Co., Seoul, South Korea) and loaded into a 1% agarose gel (BioPrince Co., Chuncheon, South Korea) containing ethidium bromide in 1% Tris-acetate EDTA (TAE) buffer (Tris-aminomethane, Glacial acetic acid, 0.5M EDTA pH 8.0). Electrophoresis was performed at 100 V for 15 min. The resulting DNA fragments were visualized and analyzed using Benchtop UV Trans illuminator (UVP, Cambridge, U.K.).
The PCR products were purified using the ExpinTM Gel SV kit (Gene-all Biotech Co., Seoul, South Korea) according to the manufacturer’s protocol. GB buffer (90 µL) was added to the PCR products and well mixed in. This mixture was then transferred to the SV column, centrifuged for 1 min at 4°C and the pass-through was discarded. NW (700 µL) buffer was added to the SV column, centrifuged for 1 min and the pass-through was likewise discarded. Residual buffer NW was dried for 5~10 min and the collection tube was transferred to a new 1.5 ml E-tube. EB buffer (20 µL) was added to the SV column, centrifuged for 1 min and purified PCR products were transferred to a new 1.5 ml E-tube. The PCR products’ sequencing was done at Macrogen (Seoul, Republic of Korea) and identified using the Basic Local Alignment Search Tool (BLAST) program. Phylogenetic tree was constructed by MEGA6 with neighbor-joining calculation method.
Soluble solids content (SSC) and Firmness measurement The SSC was measured in a fruit juice with 10 replicates. The kiwifruit was wrapped in a four-layer cheese cloth, and absorbance was read using a portable refractometer (Pocket Refractometer, PAL-1, Atago, Japan). Fruit firmness was measured with a probe diameter of 8 mm using a rheometer (RHEO TEX SD-700, Sun Scientific Inc., Japan). The kiwifruit was cut through the equator (2 cm radial slices), and firmness was measured at two points of the outer pericarp and core tissue zone by driving a depth of 3 mm at a speed of 120 mm/min. The maximum force generated during penetration was recorded as Newton (N) (McGlone and Kawano, 1998). The firmness of 10 kiwifruits was then measured.
Results and Discussion
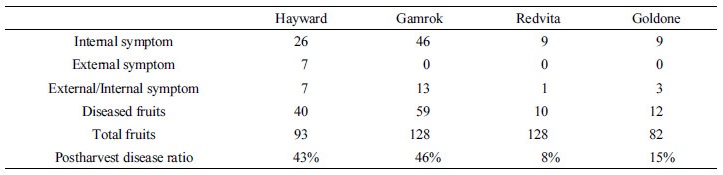
Postharvest fruit rot diseases occurred in all kiwifruit cultivars during the ripening stage. First, when four different cultivars of kiwifruit were stored at 0~1°C and ripened at 16°C for 14 days in 2014, the results were as follows. When the fruit was stored cv. Redvita and cv. Goldone were more resistant to postharvest diseases than control cv. Hayward. External symptoms appeared as dark and soft skin. All the skins of the fruit were peeled off and internal symptoms appeared as decayed flesh and dark lesions caused by water immersion (Fig. 1). Black-colored skin was seen on cv. Hayward (Fig. 1A). Green water-soaked lesions appeared on cv. Gamrok (Fig. 1B) and yellow water-soaked lesions were evident on cv. Redvita (Fig. 1C). Both external and internal symptoms were confirmed on cv. Goldone (Fig. 1D). Most disease symptoms were internal but some appeared as both external and internal symptoms. However, cultivar Hayward had only external symptoms. Postharvest diseases incidence of each cultivar was as follows: Hayward was 43%, Gamrok was 46%, Redvita was 8% and Goldone was 15% (Table 1). In comparison with existing cultivar Hayward, cv. Redvita and cv. Goldone were more resistant but cv. Gamrok was more sensitive to postharvest fruit rot disease (Table 1). Detection ratios of fungi in all cultivars were: B. dothidea was 74.38%, Phomopsis sp. was 19.83% and A. alternata was 5.79%. Here, A. alternata was isolated only from cv. Hayward (Table 2). These results indicated that yellow-fleshed new cv. Redvita and cv. Goldone are resistant to fungi of postharvest disease and safer to sell to consumers. As previously reported, B. dothidea and Phomopsis sp. are the major pathogens in kiwifruit postharvest fruit rot diseases. A. alternata was isolated only from cv. Hayward, which may suggest that the three new cultivars cab resist A. alternata following the post-ripening stage. Based on the above results, when cv. Hayward and cv. Gamrok were stored at 0~1°C for six months and forty fruits of each were sampled in each month, the results were as follows. Symptoms were the same as the above results for both cultivars. No symptoms were reported until the second (2nd month) diagnosis stage. From the third survey (3th month), disease symptoms appeared and incidence affected 3 out of 40 fruits. External symptoms appeared as soft skin-like pressed marks (Fig. 1E), internal symptoms appeared as water-soaked lesions (Fig. 1F) and both external and internal symptoms appeared as brown soft lesions (Fig. 1G). During the third stage of storage, symptoms appeared. Incidence of disease on cv. Hayward and cv. Gamrok was the same for 3 of 40 fruits (7.5%). As time passed, disease incidence of cv. Hayward was similar for 3~5 of 40 fruits but cv. Gamrok revealed a gradually increasing tendency, resulting in up to 26 of 40 fruits (65%) being affected. Between the fourth and six stages, the disease incidence of cv. Hayward revealed no significant differences compared to the early survey stages. However, cv. Gamrok did have dramatically increased disease incidence and severity.

Fig. 1.
Fungi infection symptoms of postharvest fruit rot. (A) Alternaria alternata on cv. Hayward, (B) Diaporthe sp. on cv. Gamrok, (C) Diaporthe sp. on Redvita and (D) Botryosphaeria dothidea on cv. Goldone stored at 0~1°C for one month and ripened at 16°C for 14 days. (E) External symptom, (F) internal symptoms and (G) both external and internal symptoms on cv. Gamrok stored at 0~1°C.
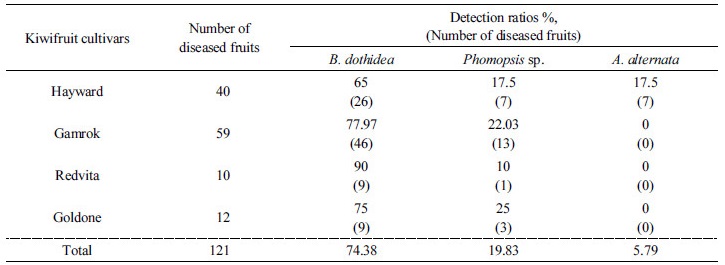
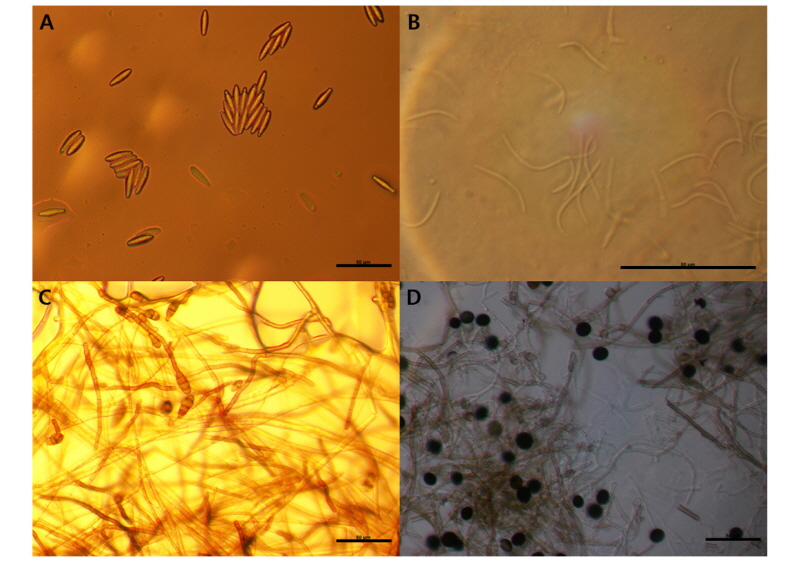
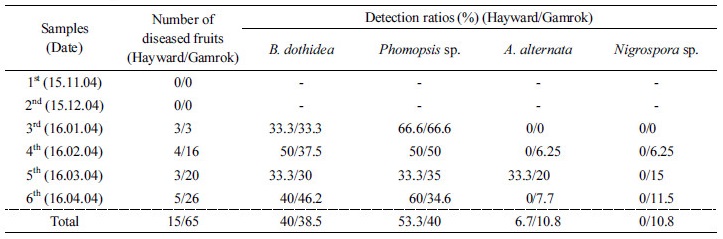
Identified fungi were B. dothidea, Phomopsis sp., A. alternata and Nigrospora sp. (Figs. 2 and 3). Total identified ratio of fungi was as follows: B. dothidea was 40%; Phomopsis sp. was 53.3%; A. alternata was 6.7% on cv. Hayward and B. dothidea was 38.5%; Phomopsis sp. was 40%; A. alternata was 10.8%; and Nigrospora sp. was 10.8% on cv. Gamrok (Table 3). Here, Nigrospora sp. was isolated only from cv. Gamrok and its detection on this particular new cultivar (Table 3). The fungi in cv. Gamrok were identified in the following order: B. dothidea, Phomopsis sp. A. alternata and Nigrospora sp. However, like cv. Hayward, B. dothidea and Phomopsis sp. were more dominant than the other fungi (Table 3). Unlike the first experiment, A. alternata was isolated in cv. Gamrok during the fourth diagnosis stage. Nigrospora sp. was isolated solely from cv. Gamrok and this particular fungus is known to infect tropical fruits such as banana, papaya and pineapple (Kader, 2002). However, until now Nigrospora sp. had not been reported in kiwifruit and this is the first documentation of it causing postharvest black rot (Kwon et al., 2017).
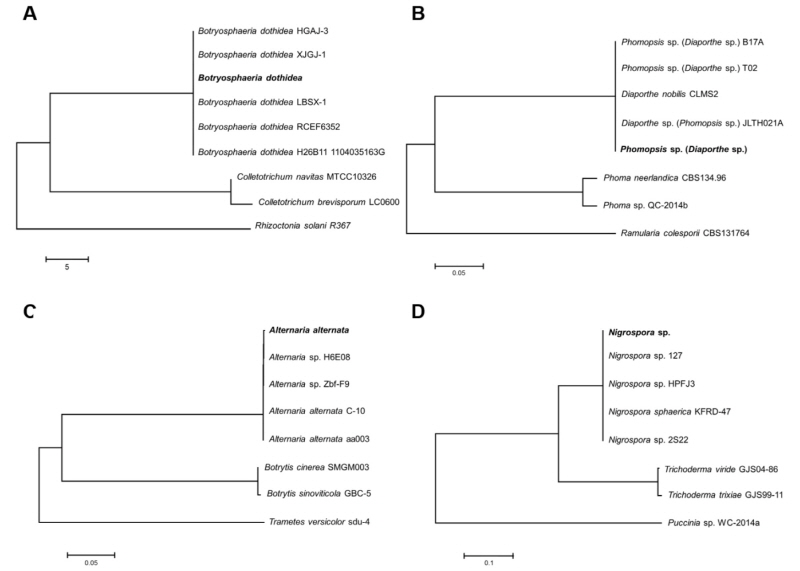
Fig. 2.
Phylogenetic identification of the fungi based on ITS sequence. (A) Botryosphaeria dothidea, (B) Phomopsis sp., (C) Alternaria alternata, and (D) Nigrospora sp. The phylogenetic tree bootstrap values are the percentage, which are calculated from 1000 replicates in a bootstrap analysis. The phylogenetic tree was calculated by the neighbor-joining method and constructed by MEGA 6.
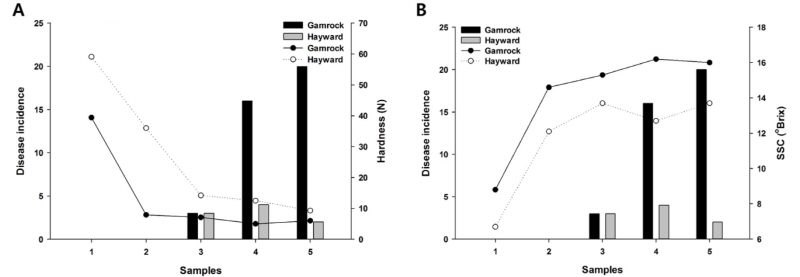
Compared with the SSC and hardness, these were significant correlations with the disease incidence. SSC and hardness had an inverse relationship, and as fruits ripened, SSC increased (Hopkirk et al., 1986) while hardness decreased (Beever and Hopkirk, 1990). The disease incidence rose when SSC was higher and hardness was lower (Fig. 4). The incidence of postharvest disease in kiwifruit may be linked to Brix degree and firmness. The higher SSC and lower firmness led to more incidence of postharvest diseases. In this case, several recommendations are considered. When kiwifruit is on sale, SSC above 12°Brix is generally considered more favorable to consumers (Stec et al., 1989). When cv. Hayward and cv. Gamrok were stored at 0~1°C for one month, SSC exceeded 12°Brix. Therefore, it would be better that both cultivars are stored at 0~1°C for one month before they are allowed to be sold in the marketplace. Another alternative is as follows. Park et al. (2006) surveyed SSC changes by treating it with ethylene. When ethylene was treated on cv. Hayward with 100µg/mL at 20°C, the SSC was significantly increased on the second day. The result suggested that with proper level of ethylene treatment to the kiwifruit immediately after the harvest season, kiwifruit can be sold to consumers free of any postharvest disease because SSC increases and acidity decreases within a few days.
In conclusion, when fruits were stored at 0~1°C for one month and ripened at 16°C for two weeks, new cultivars ‘Redvita’ and ‘Goldone’ showed more resistance to postharvest diseases than cv. Hayward. This finding suggests that the new cultivars, ‘Redvita’ and ‘Goldone’, should be given cultivation priority on South Korean kiwifruit orchards. In the case cv. Gamrok it is more sensitive to postharvest diseases than cv. Hayward, however, cv. Gamrok would enjoy better sales and public acceptance if it is first stored for one month because SSC is above 12°Brix when stored at 0~1°C for one month. Another recommendation is to treat ethylene so that sugar content increases and a post-ripening stage is not needed. The results of this study provide practical information on enhancing the quality control management of new cultivars of kiwifruit that will prevent or reduce postharvest fruit rot diseases.